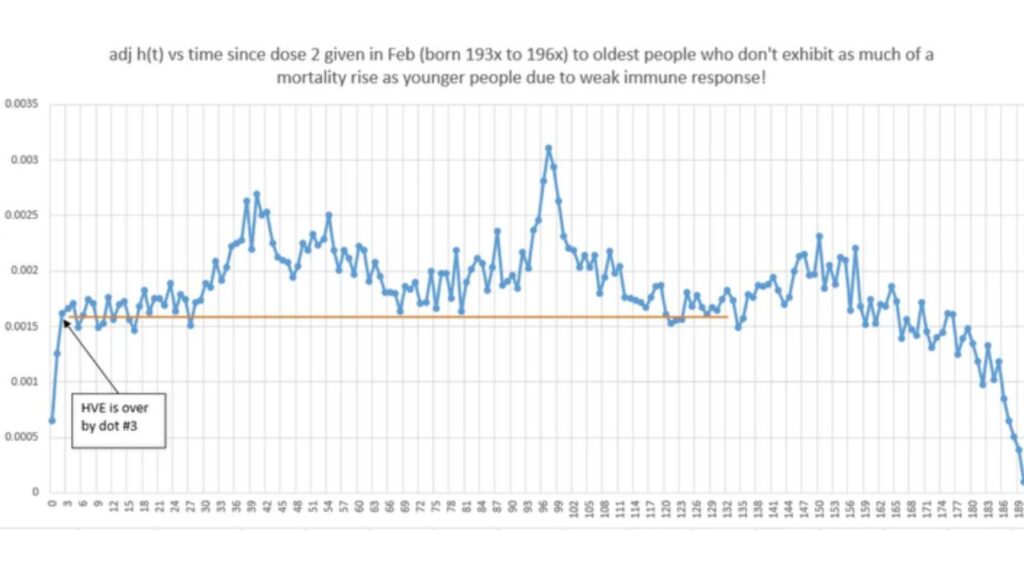
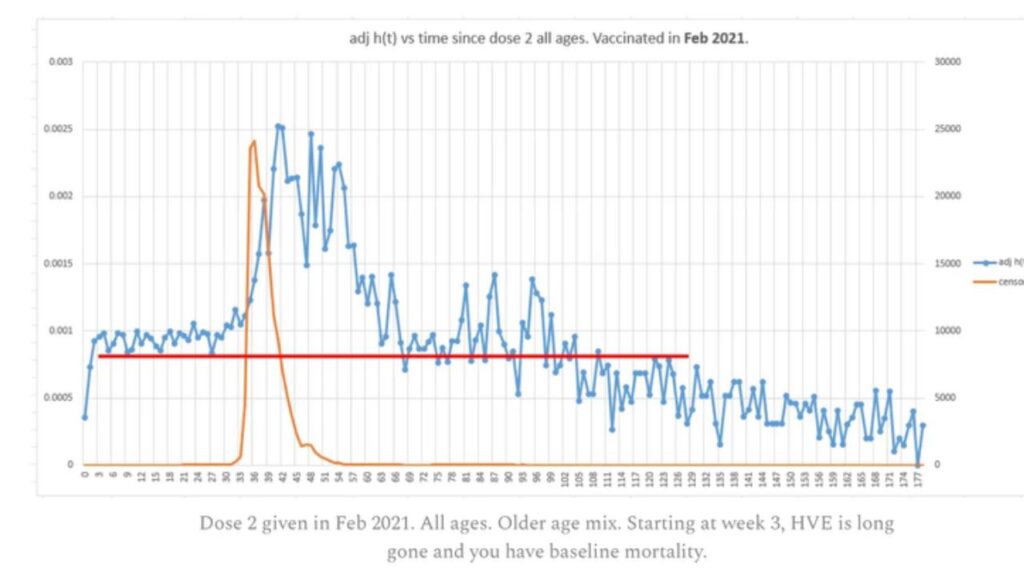
Raccolti in un solo documento 1000 studi sottoposti a revisione paritaria che mettono in dubbio la sicurezza del vaccino Covid-19
Si tratta di documenti medici sottoposti a revisione paritaria inviati a varie riviste mediche, che evidenziano una moltitudine di eventi avversi nei destinatari del vaccino Covid-19.
Si può scaricare il documento integrale qui (lo trovate in fondo alla pagina): https://web.archive.org/web/20220225041134/https://www.informedchoiceaustralia.com/post/1000-peer-reviewed-studies-questioning-covid-19-vaccine-safety
Peer Reviewed Medical Papers Submitted To Various Medical Journals, Evidencing A Multitude Of Adverse Events In Covid-19 Vaccine Recipients
- Myocarditis after mRNA vaccination against SARS-CoV-2, a case series: https://sciencedirect.com/science/article/pii/S2666602221000409
- Myocarditis after immunization with COVID-19 mRNA vaccines in members of the US military. This article reports that in “23 male patients, including 22 previously healthy military members, myocarditis wasidentified within 4 days after receipt of the vaccine”: https://jamanetwork.com/journals/jamacardiology/fullarticle/2781601
- Association of myocarditis with the BNT162b2 messenger RNA COVID-19 vaccine in a case series of children: https://pubmed.ncbi.nlm.nih.gov/34374740/
- Acute symptomatic myocarditis in seven adolescents after Pfizer-BioNTech COVID-19vaccination: https://pediatrics.aappublications.org/content/early/ 2021/06/04/peds.2021-052478
- Myocarditis and pericarditis after vaccination with COVID-19 mRNA: practical considerations for careproviders: https://sciencedirect.com/science/article/pii/ S0828282X21006243
- Myocarditis, pericarditis and cardiomyopathy after COVID-19 vaccination: https://sciencedirect.com/science/article/pii/S1443950621011562
- Myocarditis with COVID-19 mRNA vaccines: https://ahajournals.org/doi/pdf/10.1161/CIRCULATIONAHA.121.056135
- Myocarditis and pericarditis after COVID-19 vaccination: https://jamanetwork.com/journals/jama/fullarticle/2782900
- Myocarditis temporally associated with COVID-19 vaccination: https://ahajournals.org/doi/pdf/10.1161/CIRCULATIONAHA.121.055891.
- COVID-19 Vaccination Associated with Myocarditis in Adolescents: https://aappublications.org/content/pediatrics/early/2021/08/12/peds. 2021-053427.full.pdf
- Acute myocarditis after administration of BNT162b2 vaccine against COVID-19: https://pubmed.ncbi.nlm.nih.gov/33994339/
- Temporal association between COVID-19 vaccine Ad26.COV2.S and acute myocarditis: case report andreview of the literature: https://sciencedirect.com/ science/article/pii/S1553838921005789
- COVID-19 vaccine-induced myocarditis: a case report with review of the literature: https://sciencedirect.com/science/article/pii/S1871402121002253
- Potential association between COVID-19 vaccine and myocarditis: clinical and CMR findings: https://sciencedirect.com/science/article/pii/S1936878X2100485X
- Recurrence of acute myocarditis temporally associated with receipt of coronavirus mRNA disease vaccine 2019 (COVID-19) in a male adolescent: https://sciencedirect.com/science/article/pii/S002234762100617X
- Fulminant myocarditis and systemic hyper inflammation temporally associated with BNT162b2 COVID-19 mRNA vaccination in two patients: https:// sciencedirect.com/science/article/pii/S0167527321012286.
- Acute myocarditis after administration of BNT162b2 vaccine: https://sciencedirect.com/science/article/pii/S2214250921001530
- Lymphohistocytic myocarditis after vaccination with COVID-19 COV2.S viral vector: https://www.sciencedirect.com/science/article/pii/S2352906721001573
- Myocarditis following vaccination with BNT162b2 in a healthy male: https://sciencedirect.com/science/article/pii/S0735675721005362
- Acute myocarditis after Comirnaty (Pfizer) vaccination in a healthy male with previous SARS-CoV-2infection: https://sciencedirect.com/science/article/pii/ S1930043321005549
- Acute myocarditis after vaccination with SARS-CoV-2 mRNA-1273 mRNA: https://sciencedirect.com/science/article/pii/S2589790X21001931
- Acute myocarditis after SARS-CoV-2 vaccination in a 24-year-old man: https://sciencedirect.com/science/article/pii/S0870255121003243
- A series of patients with myocarditis after vaccination against SARS-CoV-2 with mRNA-1279 and BNT162b2: https://sciencedirect.com/science/article/pii/ S1936878X21004861
- COVID-19 mRNA vaccination and myocarditis: https://pubmed.ncbi.nlm.nih.gov/ 34268277/
- COVID-19 vaccine and myocarditis: https://pubmed.ncbi.nlm.nih.gov/34399967/
- Epidemiology and clinical features of myocarditis/pericarditis before the introduction of COVID-19 mRNAvaccine in Korean children: a multicenter study https:// bvsalud.org/global-literature-on-novel-coronavirus-2019-ncov/resourc e/en/ covidwho-1360706.
- COVID-19 vaccines and myocarditis: https://pubmed.ncbi.nlm.nih.gov/34246566/
- Myocarditis and other cardiovascular complications of COVID-19 mRNA-based COVID-19 vaccineshttps://cureus.com/articles/61030-myocarditis-and-other- cardiovascular-complications-of-the-mrna-based-covid-19-vaccines
- Myocarditis and other cardiovascular complications of COVID-19 mRNA-based COVID-19 vaccineshttps://cureus.com/articles/61030-myocarditis-and-other- cardiovascular-complications-of-the-mrna-based-covid-19-vaccines
- Myocarditis, pericarditis, and cardiomyopathy after COVID-19 vaccination: https://ncbi.nlm.nih.gov/34340927/
- Myocarditis with covid-19 mRNA vaccines: https://ahajournals.org/doi/10.1161/CIRCULATIONAHA.121.056135
- Association of myocarditis with COVID-19 mRNA vaccine in children: https://jamanetwork.com/news-item/association-of-myocarditis-with-mrna-co vid-19- vaccine-in-children/
- Association of myocarditis with COVID-19 messenger RNA vaccine BNT162b2 in a case series ofchildren: https://jamanetwork.com/journals/jamacardiology/fullarticle/ 2783052
- Myocarditis after immunization with COVID-19 mRNA vaccines in members of the
U.S. military: https://jamanetwork.com/journals/jamacardiology/fullarticle/ 2781601%5C
- Myocarditis occurring after immunization with COVID-19 mRNA-based COVID-19 vaccines: https://jamanetwork.com/journals/jamacardiology/fullarticle/2781600
- Myocarditis following immunization with Covid-19 mRNA: https://nejm.org/doi/full/10.1056/NEJMc2109975
- Patients with acute myocarditis after vaccination withCOVID-19 mRNA: https://com/journals/jamacardiology/fullarticle/2781602
- Myocarditis associated with vaccination with COVID-19 mRNA: https://pubs.rsna.org/doi/10.1148/radiol.2021211430
- Symptomatic Acute Myocarditis in 7 Adolescents after Pfizer-BioNTech COVID-19 Vaccination: https://pediatrics.aappublications.org/content/148/3/e2021052478
- Cardiovascular magnetic resonance imaging findings in young adult patients with acute myocarditis after COVID-19 mRNA vaccination: a case series: https://jcmr-biomedcentral.com/articles/10.1186/s12968-021-00795-4
- Clinical Guidance for Young People with Myocarditis and Pericarditis after Vaccination with COVID-19 mRNA: https://cps.ca/en/documents/position/ clinical-guidance-for-youth-with-myocarditis-and-pericarditis
- Cardiac imaging of acute myocarditis after vaccination with COVID-19 mRNA: https://ncbi.nlm.nih.gov/34402228/
- Case report: acute myocarditis after second dose of mRNA-1273 SARS-CoV-2 mRNA vaccine: https://academic.oup.com/ehjcr/article/5/8/ytab319/6339567
- Myocarditis / pericarditis associated with COVID-19 vaccine: https://science.gc.ca/eic/site/063.nsf/eng/h_98291.html
- The new COVID-19 mRNA vaccine platform and myocarditis: clues to the possible underlying mechanism: https://pubmed.ncbi.nlm.nih.gov/34312010/
- Myocarditis associated with COVID-19 vaccination: echocardiographic, cardiac tomography, and magneticresonance imaging findings: https://ahajournals.org/ doi/10.1161/CIRCIMAGING.121.013236
- In-depth evaluation of a case of presumed myocarditis after the second dose of COVID-19 mRNA vaccine: https://ahajournals.org/doi/10.1161/ CIRCULATIONAHA.121.056038
- Occurrence of acute infarct-like myocarditis after COVID-19 vaccination: just an accidental coincidence orrather a vaccination-associated autoimmune myocarditis?: https://pubmed.ncbi.nlm.nih.gov/34333695/
- Recurrence of acute myocarditis temporally associated with receipt of coronavirus mRNA disease vaccine 2019 (COVID-19) in a male adolescent: https:// ncbi.nlm.nih.gov/pmc/articles/PMC8216855/
- Myocarditis after SARS-CoV-2 vaccination: a vaccine-induced reaction?: https://ncbi.nlm.nih.gov/34118375/
- Self-limited myocarditis presenting with chest pain and ST-segment elevation in adolescents after vaccination with the BNT162b2 mRNA vaccine: https:// ncbi.nlm.nih.gov/34180390/
- Biopsy-proven lymphocytic myocarditis after first COVID-19 mRNA vaccination in a 40-year-old man: case report: https://pubmed.ncbi.nlm.nih.gov/34487236/
- Myocarditis and other cardiovascular complications of mRNA-based COVID-19 vaccines: https://pubmed.ncbi.nlm.nih.gov/34277198/
- Case report: acute myocarditis after second dose of SARS-CoV-2 mRNA-1273 vaccine mRNA-1273: https://pubmed.ncbi.nlm.nih.gov/34514306/
- Acute myocardial infarction within 24 hours after COVID-19 vaccination: https://ncbi.nlm.nih.gov/34364657/.
- fulminant myocarditis and systemic hyperinflammation temporally associated with BNT162b2 COVID-19 mRNA vaccination in two patients: https:// ncbi.nlm.nih.gov/34416319/.
- Lymphohistocytic myocarditis after vaccination with the COVID-19 viral vector Ad26.COV2.S: https://pubmed.ncbi.nlm.nih.gov/34514078/
- Myocarditis associated with SARS-CoV-2 mRNA vaccination in children aged 12 to 17 years: stratifiedanalysis of a national database: https://medrxiv.org/content/ 10.1101/2021.08.30.21262866v1
- A report of myocarditis adverse events in the S. Vaccine Adverse Event Reporting System. (VAERS) in association with COVID-19 injectable biologics: https:// pubmed.ncbi.nlm.nih.gov/34601006/
- This study concludes that: “The vaccine was associated with an excess risk of myocarditis (1 to 5 events per 100,000 persons). The risk of this potentially serious adverse event and of many other serious adverse events increased substantially after SARS-CoV-2 infection”: https://nejm.org/doi/full/10.1056/NEJMoa2110475
- Myocarditis associated with SARS-CoV-2 mRNA vaccination in children aged 12 to 17 years: stratifiedanalysis of a national database: https://medrxiv.org/content/ 10.1101/2021.08.30.21262866v1.
- Epidemiology of acute myocarditis/pericarditis in Hong Kong adolescents after co- vaccination:https://academic.oup.com/cid/advance-article-abstract/doi/10.1093/cid/ ciab989/644 5179.
- Myocarditis after 2019 coronavirus disease mRNA vaccine: a case series and determination ofincidence rate: https://academic.oup.com/cid/advance-article/doi/ 1093/cid/ciab926/6420408
- Myocarditis and pericarditis after COVID-19 vaccination: inequalities in age and vaccine types: https://mdpi.com/2075-4426/11/11/1106
- Epidemiology and clinical features of myocarditis/pericarditis before the introduction of COVID-19 mRNAvaccine in Korean children: a multicenter study: https:// ncbi.nlm.nih.gov/34402230/
- Shedding light on post-vaccination myocarditis and pericarditis in COVID-19 and non-COVID-19 vaccine recipients: https://pubmed.ncbi.nlm.nih.gov/34696294/
- Myocarditis Following mRNA COVID-19 Vaccine: https://journals.lww.com/pec-online/Abstract/2021/11000/Myocarditis_Following_ mRNA_COVID_19_Vaccine.9.aspx.
- Myocarditis following BNT162b2 mRNA Covid-19 mRNA vaccine in Israel: https://ncbi.nlm.nih.gov/34614328/.
- Myocarditis, pericarditis, and cardiomyopathy following COVID-19 vaccination: https://heartlungcirc.org/article/S1443-9506(21)01156-2/fulltext
- Myocarditis and other cardiovascular complications of COVID-19 mRNA-based COVID-19 vaccines: https://pubmed.ncbi.nlm.nih.gov/34277198/
- Possible Association Between COVID-19 Vaccine and Myocarditis: Clinical and CMR Findings: https://pubmed.ncbi.nlm.nih.gov/34246586/
- Hypersensitivity Myocarditis and COVID-19 Vaccines: https://ncbi.nlm.nih.gov/34856634/.
- Severe myocarditis associated with COVID-19 vaccine: zebra or unicorn?: https://internationaljournalofcardiology.com/article/S0167-5273(21)01477-7/fulltext.
- Acute myocardial infarction and myocarditis after COVID-19 vaccination: https://ncbi.nlm.nih.gov/labs/pmc/articles/PMC8522388/
- Myocarditis after Covid-19 vaccination in a large healthcare organization: https://nejm.org/doi/10.1056/NEJMoa2110737
- Association of myocarditis with COVID-19 messenger RNA BNT162b2 vaccine in a case series ofchildren: https://jamanetwork.com/journals/jamacardiology/fullarticle/ 2783052
- Clinical suspicion of myocarditis temporally related to COVID-19 vaccination in adolescents and young adults: https://ahajournals.org/doi/abs/10.1161/ CIRCULATIONAHA.121.056583?
url_ver=Z39.88-2003&rfr_id=ori:rid:crossref.org&rfr_dat=cr_pub%20%200pubmed
- STEMI mimicry: focal myocarditis in an adolescent patient after COVID-19 mRNA vaccination:. https://pubmed.ncbi.nlm.nih.gov/34756746/
- Myocarditis and pericarditis in association with COVID-19 mRNA vaccination: cases from a regional pharmacovigilance center: https://ncbi.nlm.nih.gov/labs/pmc/ articles/PMC8587334/
- Myocarditis after COVID-19 mRNA vaccines: https://pubmed.ncbi.nlm.nih.gov/ 34546329/.
- Patients with acute myocarditis after COVID-19 mRNA vaccination:. https://com/journals/jamacardiology/fullarticle/2781602.
- Myocarditis after COVID-19 vaccination: a case series: https://sciencedirect.com/science/article/pii/S0264410X21011725?via%3Dihub.
- Myocarditis associated with COVID-19 vaccination in adolescents: https://aap.org/pediatrics/article/148/5/e2021053427/181357
- Myocarditis findings on cardiac magnetic resonance imaging after vaccination with COVID-19 mRNA in adolescents:. https://pubmed.ncbi.nlm.nih.gov/34704459/
- Myocarditis after COVID-19 vaccination: magnetic resonance imaging study: https://oup.com/ehjcimaging/advance-article/doi/10.1093/ehjci/jeab230/6 421640.
- Acute myocarditis after administration of the second dose of BNT162b2 COVID-19 vaccine: https://ncbi.nlm.nih.gov/labs/pmc/articles/PMC8599115/
- Myocarditis after COVID-19 vaccination: https://sciencedirect.com/science/article/pii/S2352906721001603
- Case report: probable myocarditis after Covid-19 mRNA vaccine in a patient with arrhythmogenic leftventricular cardiomyopathy: https://pubmed.ncbi.nlm.nih.gov/ 34712717/.
- Acute myocarditis after administration of BNT162b2 vaccine against COVID-19: https://revespcardiol.org/en-linkresolver-acute-myocarditis-after-administration- bnt162b2-S188558572100133X.
- Myocarditis associated with COVID-19 mRNA vaccination: https://pubs.rsna.org/doi/1148/radiol.2021211430
- Acute myocarditis after COVID-19 vaccination: a case report: https://sciencedirect.com/science/article/pii/S0248866321007098
- Acute myopericarditis after COVID-19 vaccination in adolescents:. https://ncbi.nlm.nih.gov/34589238/.
- Perimyocarditis in adolescents after Pfizer-BioNTech COVID-19 vaccination: https://oup.com/jpids/article/10/10/962/6329543.
- Acute myocarditis associated with anti-COVID-19 vaccination: https://ecevr.org/ php?id=10.7774/cevr.2021.10.2.196.
- Myocarditis associated with COVID-19 vaccination: echocardiographic, cardiac CT, and MRI findings:. https://pubmed.ncbi.nlm.nih.gov/34428917/.
- Acute symptomatic myocarditis in 7 adolescents after Pfizer-BioNTech COVID-19 vaccination:. https://pubmed.ncbi.nlm.nih.gov/34088762/.
- Myocarditis and pericarditis in adolescents after first and second doses of COVID-19 mRNA vaccines:. https://academic.oup.com/ehjqcco/advance-article/doi/10.1093/ ehjqcco/qcab090/64 42104.
- COVID 19 vaccine for Concern for myocarditis and pericarditis: https:// www.mdpi.com/2036-7503/13/3/61.
- Cardiac imaging of acute myocarditis after vaccination with COVID-19 mRNA: https://ncbi.nlm.nih.gov/34402228/
- Myocarditis temporally associated with COVID-19 vaccination: https://ncbi.nlm.nih.gov/34133885/
- Acute myocarditis associated with COVID-19 vaccination: report of a case: https://ncbi.nlm.nih.gov/labs/pmc/articles/PMC8639400/
- Myocarditis following vaccination with COVID-19 messenger RNA: a Japanese case series: https://pubmed.ncbi.nlm.nih.gov/34840235/.
- Myocarditis in the setting of a recent COVID-19 vaccination: https://ncbi.nlm.nih.gov/34712497/.
- Acute myocarditis after a second dose of COVID-19 mRNA vaccine: report of two cases: https://clinicalimaging.org/article/S0899-7071(21)00265-5/fulltext.
- Prevalence of thrombocytopenia, antiplatelet factor 4 antibodies, and elevated D- dimer in Thais after vaccination with ChAdOx1 nCoV-19: https:// ncbi.nlm.nih.gov/34568726/
- Epidemiology of acute myocarditis/pericarditis in Hong Kong adolescents after co- vaccination:https://academic.oup.com/cid/advance-article-abstract/doi/10.1093/cid/ ciab989/6445179
- Myocarditis after 2019 coronavirus disease mRNA vaccine: a case series and incidence ratedetermination: https://academic.oup.com/cid/advance-article/doi/ 1093/cid/ciab926/6420408.
- Myocarditis and pericarditis after COVID-19 vaccination: inequalities in age and vaccine types: https://mdpi.com/2075-4426/11/11/1106
- Epidemiology and clinical features of myocarditis/pericarditis before the introduction of COVID-19 mRNAvaccine in Korean children: a multicenter study: https:// ncbi.nlm.nih.gov/34402230/
- Shedding light on post-vaccination myocarditis and pericarditis in COVID-19 and non-COVID-19 vaccine recipients: https://pubmed.ncbi.nlm.nih.gov/34696294/
- Epidemiology of acute myocarditis/pericarditis in Hong Kong adolescents after co- vaccination: https://pubmed.ncbi.nlm.nih.gov/34849657/.
- Myocarditis-induced sudden death after BNT162b2 COVID-19 mRNA vaccination in Korea: case report focusing on histopathological findings: https:// ncbi.nlm.nih.gov/34664804/
- Acute myocarditis after vaccination with COVID-19 mRNA in adults aged 18 years or older: https://pubmed.ncbi.nlm.nih.gov/34605853/
- Recurrence of acute myocarditis temporally associated with receipt of the 2019 coronavirus mRNAdisease vaccine (COVID-19) in an adolescent male: https:// ncbi.nlm.nih.gov/34166671/
- Young male with myocarditis after mRNA-1273 coronavirus disease-2019 (COVID-19) mRNAvaccination: https://pubmed.ncbi.nlm.nih.gov/34744118/
- Acute myocarditis after SARS-CoV-2 vaccination in a 24-year-old male: https://ncbi.nlm.nih.gov/34334935/.
- Ga-DOTATOC digital PET images of inflammatory cell infiltrates in myocarditis after vaccination with COVID-19: https://pubmed.ncbi.nlm.nih.gov/34746968/
- Occurrence of acute infarct-like myocarditis after vaccination with COVID-19: just an accidental coincidenceor rather a vaccination-associated autoimmune myocarditis?”: https://pubmed.ncbi.nlm.nih.gov/34333695/.
- Self-limited myocarditis presenting with chest pain and ST-segment elevation in adolescents after vaccination with BNT162b2 mRNA vaccine: https:// ncbi.nlm.nih.gov/34180390/
- Myocarditis Following Immunization with COVID-19 mRNA Vaccines in Members of the U.S. Military: https://pubmed.ncbi.nlm.nih.gov/34185045/
- Myocarditis after BNT162b2 vaccination in a healthy male: https://ncbi.nlm.nih.gov/34229940/
- Acute myocarditis after SARS-CoV-2 mRNA-1273 mRNA vaccination: https://ncbi.nlm.nih.gov/34308326/.
- Biopsy-proven lymphocytic myocarditis after first vaccination with COVID-19 mRNA in a 40-year-old man: case report: https://pubmed.ncbi.nlm.nih.gov/34487236/
- Multimodality imaging and histopathology in a young man presenting with fulminant lymphocytic myocarditis and cardiogenic shock after vaccination with mRNA-1273:https://pubmed.ncbi.nlm.nih.gov/34848416/
- Acute myocarditis after Comirnaty vaccination in a healthy male with previous SARS-CoV-2 infection: https://pubmed.ncbi.nlm.nih.gov/34367386/
- Acute myocarditis in a young adult two days after vaccination with Pfizer: https://ncbi.nlm.nih.gov/34709227/
- Case report: acute fulminant myocarditis and cardiogenic shock after messenger RNA coronavirus vaccination in 2019 requiring extracorporeal cardiopulmonary resuscitation: https://pubmed.ncbi.nlm.nih.gov/34778411/
- Acute myocarditis after 2019 coronavirus disease vaccination: https://ncbi.nlm.nih.gov/34734821/
- A series of patients with myocarditis after vaccination against SARS-CoV-2 with mRNA-1279 and BNT162b2: https://pubmed.ncbi.nlm.nih.gov/34246585/
- Acute myocarditis defined after vaccination with 2019 mRNA of coronavirus disease: https://pubmed.ncbi.nlm.nih.gov/34866122/
- Biventricular systolic dysfunction in acute myocarditis after SARS-CoV-2 mRNA-1273 vaccination: https://pubmed.ncbi.nlm.nih.gov/34601566/
- Myocarditis following COVID-19 vaccination: MRI study: https://ncbi.nlm.nih.gov/34739045/.
- Acute myocarditis after COVID-19 vaccination: case report: https://docs.google.com/document/d/1Hc4bh_qNbZ7UVm5BLxkRdMPnnI9zcCsl/e
- Association of myocarditis with COVID-19 messenger RNA BNT162b2 vaccine COVID-19 in a caseseries of children: https://pubmed.ncbi.nlm.nih.gov/34374740/
- Clinical suspicion of myocarditis temporally related to COVID-19 vaccination in adolescents and young adults: https://pubmed.ncbi.nlm.nih.gov/34865500/
- Myocarditis following vaccination with Covid-19 in a large healthcare organization: https://pubmed.ncbi.nlm.nih.gov/34614329/
- Myocarditis and pericarditis in adolescents after the first and second doses of COVID-19 mRNA vaccines: https://pubmed.ncbi.nlm.nih.gov/34849667/
- Myocarditis after SARS-CoV-2 mRNA vaccination, a case series: https://ncbi.nlm.nih.gov/34396358/.
- Epidemiology of myocarditis and pericarditis following mRNA vaccines in Ontario, Canada: by vaccineproduct, schedule, and interval: https://medrxiv.org/content/ 10.1101/2021.12.02.21267156v1
- Acute myocardial infarction and myocarditis after COVID-19 vaccination: https://ncbi.nlm.nih.gov/34586408/
- Abbate, , Gavin, J., Madanchi, N., Kim, C., Shah, P. R., Klein, K., . . . Danielides,
- (2021). Fulminant myocarditis and systemic hyperinflammation temporally associated with BNT162b2mRNA COVID-19 vaccination in two patients. Int J Cardiol, 340, 119-121. doi:10.1016/j.ijcard.2021.08.018. https:// www.ncbi.nlm.nih.gov/pubmed/34416319
- Abu Mouch, S., Roguin, , Hellou, E., Ishai, A., Shoshan, U., Mahamid, L., . . . Berar Yanay, N. (2021).Myocarditis following COVID-19 mRNA vaccination. Vaccine,
39(29), 3790-3793. doi:10.1016/j.vaccine.2021.05.087. https://www.ncbi.nlm.nih.gov/ pubmed/34092429
- Albert, E., Aurigemma, G., Saucedo, J., & Gerson, D. S. (2021). Myocarditis following COVID-19 Radiol Case Rep, 16(8), 2142-2145. doi:10.1016/ j.radcr.2021.05.033. https://www.ncbi.nlm.nih.gov/pubmed/34025885
- Aye, N., Mai, A. S., Zhang, A., Lim, O. Z. H., Lin, N., Ng, C. H., . . . Chew, N. W. S.
(2021). Acute Myocardial Infarction and Myocarditis following COVID-19 Vaccination. QJM. doi:10.1093/qjmed/hcab252. https://www.ncbi.nlm.nih.gov/pubmed/34586408
- Azir, M., Inman, B., Webb, J., & Tannenbaum, L. (2021). STEMI Mimic: Focal Myocarditis in anAdolescent Patient After mRNA COVID-19 J Emerg Med, 61(6), e129-e132. doi:10.1016/j.jemermed.2021.09.017. https:// www.ncbi.nlm.nih.gov/pubmed/34756746
- Bozkurt, B., Kamat, I., & Hotez, P. J. (2021). Myocarditis With COVID-19 mRNA Vaccines. Circulation,144(6), 471-484. doi:10.1161/CIRCULATIONAHA.121.056135.
https://www.ncbi.nlm.nih.gov/pubmed/34281357
- Calcaterra, , Mehta, J. L., de Gregorio, C., Butera, G., Neroni, P., Fanos, V., & Bassareo, P. P. (2021). COVID 19 Vaccine for Adolescents. Concern about Myocarditis and Pericarditis. Pediatr Rep, 13(3), 530-533. doi:10.3390/ pediatric13030061. https://www.ncbi.nlm.nih.gov/pubmed/34564344
- Chamling, , Vehof, V., Drakos, S., Weil, M., Stalling, P., Vahlhaus, C., . . . Yilmaz,
- (2021). Occurrence of acute infarct-like myocarditis following COVID-19 vaccination: just an accidental co-incidence or rather vaccination-associated autoimmune myocarditis? Clin Res Cardiol,110(11), 1850-1854. doi:10.1007/ s00392-021-01916-w. https://www.ncbi.nlm.nih.gov/pubmed/34333695
- Chelala, , Jeudy, J., Hossain, R., Rosenthal, G., Pietris, N., & White, C. (2021). Cardiac MRI Findings of Myocarditis After COVID-19 mRNA Vaccination in Adolescents. AJR Am J Roentgenol. doi:10.2214/AJR.21.26853. https:// www.ncbi.nlm.nih.gov/pubmed/34704459
- Choi, , Lee, S., Seo, J. W., Kim, M. J., Jeon, Y. H., Park, J. H., . . . Yeo, N. S.
(2021). Myocarditis-induced Sudden Death after BNT162b2 mRNA COVID-19 Vaccination in Korea: CaseReport Focusing on Histopathological Findings. J Korean Med Sci, 36(40), e286. doi:10.3346/jkms.2021.36.e286. https:// www.ncbi.nlm.nih.gov/pubmed/34664804
- Chua, T., Kwan, M. Y. W., Chui, C. S. L., Smith, R. D., Cheung, E. C., Tian, T., . . .
Ip, P. (2021). Epidemiology of Acute Myocarditis/Pericarditis in Hong Kong Adolescents FollowingComirnaty Vaccination. Clin Infect Dis. doi:10.1093/cid/ ciab989. https://www.ncbi.nlm.nih.gov/pubmed/34849657
- Clarke, , & Ioannou, A. (2021). Should T2 mapping be used in cases of recurrent myocarditis to differentiate between the acute inflammation and chronic scar? J Pediatr. doi:10.1016/j.jpeds.2021.12.026. https://www.ncbi.nlm.nih.gov/pubmed/ 34933012
- Das, B. B., Moskowitz, W. B., Taylor, M. B., & Palmer, (2021). Myocarditis and Pericarditis Following mRNA COVID-19 Vaccination: What Do We Know So Far? Children (Basel), 8(7).doi:10.3390/children8070607. https://www.ncbi.nlm.nih.gov/ pubmed/34356586
- Dickey, J. B., Albert, E., Badr, M., Laraja, K. M., Sena, L. M., Gerson, D. S., . . . Aurigemma, P.(2021). A Series of Patients With Myocarditis Following SARS- CoV-2 Vaccination With mRNA-1279 andBNT162b2. JACC Cardiovasc Imaging, 14(9), 1862-1863. doi:10.1016/j.jcmg.2021.06.003.https://www.ncbi.nlm.nih.gov/ pubmed/34246585
- Ehrlich, P., Klingel, K., Ohlmann-Knafo, S., Huttinger, S., Sood, N., Pickuth, D., & Kindermann, (2021).Biopsy-proven lymphocytic myocarditis following first mRNA COVID-19 vaccination in a 40-year-old male: case report. Clin Res Cardiol, 110(11), 1855-1859. doi:10.1007/s00392-021-01936-6.https://www.ncbi.nlm.nih.gov/pubmed/
34487236
- Facetti, S., Giraldi, M., Vecchi, L., Rogiani, S., & Nassiacos, D. (2021). [Acute myocarditis in a youngadult two days after Pfizer vaccination]. G Ital Cardiol (Rome), 22(11), 891-893. doi:10.1714/3689.36746. https://www.ncbi.nlm.nih.gov/pubmed/
34709227
- Foltran, D., Delmas, C., Flumian, C., De Paoli, P., Salvo, F., Gautier, S., . . . Montastruc, F. (2021). Myocarditis and Pericarditis in Adolescents after First and Second doses of mRNA COVID-19 EurHeart J Qual Care Clin Outcomes. doi:10.1093/ehjqcco/qcab090. https://www.ncbi.nlm.nih.gov/pubmed/34849667
- Gargano, W., Wallace, M., Hadler, S. C., Langley, G., Su, J. R., Oster, M. E., . . . Oliver, S. E. (2021). Use of mRNA COVID-19 Vaccine After Reports of Myocarditis Among Vaccine Recipients: Update from theAdvisory Committee on Immunization Practices – United States, June 2021. MMWR Morb Mortal Wkly Rep, 70(27),
977-982. doi:10.15585/mmwr.mm7027e2. https://www.ncbi.nlm.nih.gov/pubmed/
34237049
- Gautam, , Saluja, P., Fudim, M., Jambhekar, K., Pandey, T., & Al’Aref, S. (2021). A Late Presentation ofCOVID-19 Vaccine-Induced Myocarditis. Cureus, 13(9), e17890. doi:10.7759/cureus.17890. https://www.ncbi.nlm.nih.gov/pubmed/34660088
- Gellad, F. (2021). Myocarditis after vaccination against covid-19. BMJ, 375, n3090. doi:10.1136/bmj.n3090. https://www.ncbi.nlm.nih.gov/pubmed/34916217
- In brief: Myocarditis with the Pfizer/BioNTech and Moderna COVID-19 (2021). Med Lett Drugs Ther, 63(1629), e9. Retrieved from https://www.ncbi.nlm.nih.gov/pubmed/34544112https://www.ncbi.nlm.nih.gov/pubmed/ 3454412
- Ioannou, (2021a). Myocarditis should be considered in those with a troponin rise and unobstructedcoronary arteries following Pfizer-BioNTech COVID-19 vaccination. QJM. doi:10.1093/qjmed/hcab231. https://www.ncbi.nlm.nih.gov/pubmed/34463755
- Ioannou, (2021b). T2 mapping should be utilised in cases of suspected myocarditis to confirm anacute inflammatory process. QJM. doi:10.1093/qjmed/ hcab326. https://www.ncbi.nlm.nih.gov/pubmed/34931681
- Isaak, , Feisst, A., & Luetkens, J. A. (2021). Myocarditis Following COVID-19 Vaccination. Radiology,301(1), E378-E379. doi:10.1148/radiol.2021211766. https:// www.ncbi.nlm.nih.gov/pubmed/34342500
- Istampoulouoglou, I., Dimitriou, G., Spani, S., Christ, A., Zimmermanns, B., Koechlin, , . . . Leuppi-Taegtmeyer, A. B. (2021). Myocarditis and pericarditis in association with COVID-19 mRNA-vaccination: cases from a regional
pharmacovigilance centre. Glob Cardiol Sci Pract, 2021(3), e202118. doi:10.21542/ gcsp.2021.18. https://www.ncbi.nlm.nih.gov/pubmed/34805376
- Jain, S., Steele, J. M., Fonseca, B., Huang, S., Shah, S., Maskatia, S. A., . . . Grosse-Wortmann, L. (2021). COVID-19 Vaccination-Associated Myocarditis in Adolescents. Pediatrics, 148(5). doi:10.1542/peds.2021-053427. https:// www.ncbi.nlm.nih.gov/pubmed/34389692
- Kaneta, , Yokoi, K., Jojima, K., Kotooka, N., & Node, K. (2021). Young Male With Myocarditis Following mRNA-1273 Vaccination Against Coronavirus Disease-2019 (COVID-19). Circ J. doi:10.1253/circj.CJ-21-0818. https://www.ncbi.nlm.nih.gov/ pubmed/34744118
- Kaul, , Sreenivasan, J., Goel, A., Malik, A., Bandyopadhyay, D., Jin, C., . . . Panza,
- A. (2021). Myocarditis following COVID-19 vaccination. Int J Cardiol Heart Vasc, 36, 100872.doi:10.1016/j.ijcha.2021.100872. https://www.ncbi.nlm.nih.gov/pubmed/
34568540
- Kim, W., Jenista, E. R., Wendell, D. C., Azevedo, C. F., Campbell, M. J., Darty, S. N., . . . Kim, R. J. (2021). Patients With Acute Myocarditis Following mRNA
COVID-19 Vaccination. JAMA Cardiol, 6(10), 1196-1201. doi:10.1001/jamacardio. 2021.2828. https://www.ncbi.nlm.nih.gov/pubmed/34185046
- Kim, I. C., Kim, H., Lee, H. J., Kim, J. Y., & Kim, J. Y. (2021). Cardiac Imaging of Acute Myocarditis Following COVID-19 mRNA J Korean Med Sci, 36(32), e229. doi:10.3346/jkms.2021.36.e229.https://www.ncbi.nlm.nih.gov/pubmed/ 34402228
- King, W. W., Petersen, M. R., Matar, R. M., Budweg, J. B., Cuervo Pardo, L., & Petersen, J. W. (2021). Myocarditis following mRNA vaccination against SARS- CoV-2, a case Am Heart J Plus, 8, 100042.doi:10.1016/j.ahjo.2021.100042. https://www.ncbi.nlm.nih.gov/pubmed/34396358
- Kwan, Y. W., Chua, G. T., Chow, C. B., Tsao, S. S. L., To, K. K. W., Yuen, K. Y., . .
. Ip, P. (2021). mRNA COVID vaccine and myocarditis in adolescents. Hong Kong Med J, 27(5), 326-327. doi:10.12809/hkmj215120. https://www.ncbi.nlm.nih.gov/ pubmed/34393110
- Lee, , Chew, N. W. S., Ng, P., & Yeo, T. J. (2021). Reply to “Letter to the editor: Myocarditis should be considered in those with a troponin rise and unobstructed coronary arteries following PfizerBioNTech COVID-19 vaccination”. QJM. doi: 10.1093/qjmed/hcab232. https://www.ncbi.nlm.nih.gov/pubmed/34463770
- Levin, , Shimon, G., Fadlon-Derai, M., Gershovitz, L., Shovali, A., Sebbag, A., . . . Gordon, B. (2021). Myocarditis following COVID-19 vaccination – A case series. Vaccine, 39(42), 6195-6200. doi:10.1016/j.vaccine.2021.09.004. https:// www.ncbi.nlm.nih.gov/pubmed/34535317
- Li, M., Yuan, J., Lv, G., Brown, J., Jiang, X., & Lu, Z. K. (2021). Myocarditis and Pericarditis following COVID-19 Vaccination: Inequalities in Age and Vaccine J Pers Med, 11(11). doi:10.3390/jpm11111106. https://www.ncbi.nlm.nih.gov/pubmed/ 34834458
- Lim, , Kim, M. C., Kim, K. H., Jeong, I. S., Cho, Y. S., Choi, Y. D., & Lee, J. E.
(2021). Case Report: Acute Fulminant Myocarditis and Cardiogenic Shock After Messenger RNA CoronavirusDisease 2019 Vaccination Requiring Extracorporeal
Cardiopulmonary Resuscitation. Front Cardiovasc Med, 8, 758996. doi:10.3389/ fcvm.2021.758996. https://www.ncbi.nlm.nih.gov/pubmed/34778411
- Luk, , Clarke, B., Dahdah, N., Ducharme, A., Krahn, A., McCrindle, B., . . . McDonald, M. (2021). Myocarditis and Pericarditis After COVID-19 mRNA Vaccination: Practical Considerations for Care Providers. Can J Cardiol, 37(10), 1629-1634. doi:10.1016/j.cjca.2021.08.001.https://www.ncbi.nlm.nih.gov/pubmed/
34375696
- Mevorach, , Anis, E., Cedar, N., Bromberg, M., Haas, E. J., Nadir, E., . . . Alroy- Preis, S. (2021). Myocarditis after BNT162b2 mRNA Vaccine against Covid-19 in Israel. N Engl J Med, 385(23), 2140-2149. doi:10.1056/NEJMoa2109730. https:// www.ncbi.nlm.nih.gov/pubmed/34614328
- Minocha, P. K., Better, D., Singh, R. K., & Hoque, T. (2021). Recurrence of Acute Myocarditis TemporallyAssociated with Receipt of the mRNA Coronavirus Disease 2019 (COVID-19) Vaccine in a Male J Pediatr, 238, 321-323. doi: 10.1016/j.jpeds.2021.06.035. https://www.ncbi.nlm.nih.gov/pubmed/34166671
- Montgomery, , Ryan, M., Engler, R., Hoffman, D., McClenathan, B., Collins, L., . . . Cooper, L. T., Jr.(2021). Myocarditis Following Immunization With mRNA COVID-19 Vaccines in Members of the US Military. JAMA Cardiol, 6(10), 1202-1206. doi: 10.1001/jamacardio.2021.2833. https://www.ncbi.nlm.nih.gov/pubmed/34185045
- Murakami, , Shinohara, M., Oka, Y., Wada, R., Noike, R., Ohara, H., . . . Ikeda, T. (2021). MyocarditisFollowing a COVID-19 Messenger RNA Vaccination: A Japanese Case Series. Intern Med. doi:10.2169/internalmedicine.8731-21. https:// www.ncbi.nlm.nih.gov/pubmed/34840235
- Nagasaka, , Koitabashi, N., Ishibashi, Y., Aihara, K., Takama, N., Ohyama, Y., . . . Kaneko, Y. (2021).Acute Myocarditis Associated with COVID-19 Vaccination: A Case Report. J Cardiol Cases. doi:10.1016/j.jccase.2021.11.006. https:// www.ncbi.nlm.nih.gov/pubmed/34876937
- Park, , Yun, K. W., Kim, K. R., Song, S. H., Ahn, B., Kim, D. R., . . . Kim, Y. J.
(2021). Epidemiology and Clinical Features of Myocarditis/Pericarditis before the Introduction of mRNACOVID-19 Vaccine in Korean Children: a Multicenter Study. J Korean Med Sci, 36(32), e232. doi:10.3346/jkms.2021.36.e232. https:// www.ncbi.nlm.nih.gov/pubmed/34402230
- Park, , Brekke, D. R., & Bratincsak, A. (2021). Self-limited myocarditis presenting with chest pain and ST segment elevation in adolescents after vaccination with the BNT162b2 mRNA vaccine. Cardiol Young, 1-4. doi:10.1017/S1047951121002547. https://www.ncbi.nlm.nih.gov/pubmed/34180390
- Patel, Y. R., Louis, D. W., Atalay, M., Agarwal, S., & Shah, N. R. (2021). Cardiovascular magnetic resonance findings in young adult patients with acute myocarditis following mRNA COVID-19vaccination: a case J Cardiovasc Magn Reson, 23(1), 101. doi:10.1186/s12968-021-00795-4. https:// www.ncbi.nlm.nih.gov/pubmed/34496880
- Patone, M., Mei, X. W., Handunnetthi, L., Dixon, S., Zaccardi, F., Shankar-Hari, M., . . . Hippisley-Cox, J. (2021). Risks of myocarditis, pericarditis, and cardiac arrhythmias associated with COVID-19vaccination or SARS-CoV-2 Nat Med. doi:10.1038/s41591-021-01630-0. https://www.ncbi.nlm.nih.gov/pubmed/ 34907393
- Patrignani, , Schicchi, N., Calcagnoli, F., Falchetti, E., Ciampani, N., Argalia, G., & Mariani, A. (2021).Acute myocarditis following Comirnaty vaccination in a healthy man with previous SARS-CoV-2 infection.Radiol Case Rep, 16(11), 3321-3325. doi: 10.1016/j.radcr.2021.07.082. https://www.ncbi.nlm.nih.gov/pubmed/34367386
- Perez, , Levy, E. R., Joshi, A. Y., Virk, A., Rodriguez-Porcel, M., Johnson, M., . . . Swift, M. D. (2021).Myocarditis Following COVID-19 mRNA Vaccine: A Case Series and Incidence Rate Determination. Clin Infect Dis. doi:10.1093/cid/ciab926. https:// www.ncbi.nlm.nih.gov/pubmed/34734240
- Shiyovich, , Witberg, G., Aviv, Y., Eisen, A., Orvin, K., Wiessman, M., . . . Hamdan,
- (2021). Myocarditis following COVID-19 vaccination: magnetic resonance imaging study. Eur Heart J Cardiovasc Imaging. doi:10.1093/ehjci/jeab230. https:// www.ncbi.nlm.nih.gov/pubmed/34739045
- Simone, , Herald, J., Chen, A., Gulati, N., Shen, A. Y., Lewin, B., & Lee, M. S. (2021). Acute MyocarditisFollowing COVID-19 mRNA Vaccination in Adults Aged 18 Years or Older. JAMA Intern Med, 181(12), 1668-1670. doi:10.1001/jamainternmed. 2021.5511. https://www.ncbi.nlm.nih.gov/pubmed/34605853
- Singer, E., Taub, I. B., & Kaelber, D. C. (2021). Risk of Myocarditis from
COVID-19 Infection in People Under Age 20: A Population-Based Analysis. medRxiv. doi:10.1101/2021.07.23.21260998. https://www.ncbi.nlm.nih.gov/pubmed/34341797
- Starekova, J., Bluemke, D. , Bradham, W. S., Grist, T. M., Schiebler, M. L., & Reeder, S. B. (2021).Myocarditis Associated with mRNA COVID-19 Vaccination. Radiology, 301(2), E409-E411. doi:10.1148/radiol.2021211430. https:// www.ncbi.nlm.nih.gov/pubmed/34282971
- Sulemankhil, I., Abdelrahman, M., & Negi, S. I. (2021). Temporal association between the COVID-19COV2.S vaccine and acute myocarditis: A case report and literature review. Cardiovasc Revasc Med. doi:10.1016/j.carrev.2021.08.012. https://www.ncbi.nlm.nih.gov/pubmed/34420869
- Tailor, D., Feighery, A. M., El-Sabawi, B., & Prasad, A. (2021). Case report: acute myocarditis following the second dose of mRNA-1273 SARS-CoV-2 vaccine. Eur Heart J Case Rep, 5(8), ytab319. doi:10.1093/ehjcr/ytab319. https:// www.ncbi.nlm.nih.gov/pubmed/34514306
- Takeda, , Ishio, N., Shoji, T., Mori, N., Matsumoto, M., & Shikama, N. (2021). Eosinophilic Myocarditis Following Coronavirus Disease 2019 (COVID-19) Vaccination. Circ J. doi:10.1253/circj.CJ-21-0935. https://www.ncbi.nlm.nih.gov/ pubmed/34955479
- Truong, T., Dionne, A., Muniz, J. C., McHugh, K. E., Portman, M. A., Lambert, L. M., . . . Newburger, J. W. (2021). Clinically Suspected Myocarditis Temporally Related to COVID-19 Vaccination in Adolescentsand Young Adults. Circulation. doi: 10.1161/CIRCULATIONAHA.121.056583. https://www.ncbi.nlm.nih.gov/pubmed/ 34865500
- Vidula, M. K., Ambrose, M., Glassberg, H., Chokshi, N., Chen, , Ferrari, V. A., & Han, Y. (2021).Myocarditis and Other Cardiovascular Complications of the mRNA- Based COVID-19 Vaccines. Cureus, 13(6), e15576. doi:10.7759/cureus.15576. https://www.ncbi.nlm.nih.gov/pubmed/34277198
- Visclosky, T., Theyyunni, N., Klekowski, N., & Bradin, S. (2021). Myocarditis Following mRNA COVID-19 Vaccine. Pediatr Emerg Care, 37(11), 583-584. doi: 1097/PEC.0000000000002557. https://www.ncbi.nlm.nih.gov/pubmed/34731877
- Watkins, K., Griffin, G., Septaric, K., & Simon, E. L. (2021). Myocarditis after BNT162b2 vaccination in ahealthy Am J Emerg Med, 50, 815 e811-815 e812. doi:10.1016/j.ajem.2021.06.051. https://www.ncbi.nlm.nih.gov/pubmed/34229940
- Witberg, , Barda, N., Hoss, S., Richter, I., Wiessman, M., Aviv, Y., . . . Kornowski,
- (2021). Myocarditis after Covid-19 Vaccination in a Large Health Care Organization. N Engl J Med,385(23), 2132-2139. doi:10.1056/NEJMoa2110737. https://www.ncbi.nlm.nih.gov/pubmed/34614329
- In brief: Myocarditis with the Pfizer/BioNTech and Moderna COVID-19 (2021). Med Lett Drugs Ther, 63(1629), e9. Retrieved from https://www.ncbi.nlm.nih.gov/pubmed/34544112https://www.ncbi.nlm.nih.gov/pubmed/ 3454412
- Ioannou, (2021a). Myocarditis should be considered in those with a troponin rise and unobstructedcoronary arteries following Pfizer-BioNTech COVID-19 vaccination. QJM. doi:10.1093/qjmed/hcab231. https://www.ncbi.nlm.nih.gov/pubmed/34463755
- Isaak, , Feisst, A., & Luetkens, J. A. (2021). Myocarditis Following COVID-19 Vaccination. Radiology,301(1), E378-E379. doi:10.1148/radiol.2021211766. https:// www.ncbi.nlm.nih.gov/pubmed/34342500
- Istampoulouoglou, I., Dimitriou, G., Spani, S., Christ, A., Zimmermanns, B., Koechlin, S., . . . Leuppi-Taegtmeyer, B. (2021). Myocarditis and pericarditis in association with COVID-19 mRNA-vaccination: cases from a regional pharmacovigilance centre. Glob Cardiol Sci Pract, 2021(3), e202118. doi:10.21542/ gcsp.2021.18. https://www.ncbi.nlm.nih.gov/pubmed/34805376
- Jain, S., Steele, J. M., Fonseca, B., Huang, S., Shah, S., Maskatia, S. A., . . . Grosse-Wortmann, L. (2021). COVID-19 Vaccination-Associated Myocarditis in Adolescents. Pediatrics, 148(5). doi:10.1542/peds.2021-053427. https:// www.ncbi.nlm.nih.gov/pubmed/34389692
- Kaneta, , Yokoi, K., Jojima, K., Kotooka, N., & Node, K. (2021). Young Male With Myocarditis Following mRNA-1273 Vaccination Against Coronavirus Disease-2019 (COVID-19). Circ J. doi:10.1253/circj.CJ-21-0818. https://www.ncbi.nlm.nih.gov/ pubmed/34744118
- Kaul, , Sreenivasan, J., Goel, A., Malik, A., Bandyopadhyay, D., Jin, C., . . . Panza,
- A. (2021). Myocarditis following COVID-19 vaccination. Int J Cardiol Heart Vasc, 36, 100872.doi:10.1016/j.ijcha.2021.100872. https://www.ncbi.nlm.nih.gov/pubmed/
34568540
- Kim, W., Jenista, E. R., Wendell, D. C., Azevedo, C. F., Campbell, M. J., Darty, S. N., . . . Kim, R. J. (2021). Patients With Acute Myocarditis Following mRNA
COVID-19 Vaccination. JAMA Cardiol, 6(10), 1196-1201. doi:10.1001/jamacardio.
2021.2828. https://www.ncbi.nlm.nih.gov/pubmed/34185046
- Kim, I. C., Kim, H., Lee, H. J., Kim, J. Y., & Kim, J. Y. (2021). Cardiac Imaging of Acute Myocarditis Following COVID-19 mRNA J Korean Med Sci, 36(32), e229. doi:10.3346/jkms.2021.36.e229.https://www.ncbi.nlm.nih.gov/pubmed/ 34402228
- King, W. W., Petersen, M. R., Matar, R. M., Budweg, J. B., Cuervo Pardo, L., & Petersen, J. W. (2021). Myocarditis following mRNA vaccination against SARS- CoV-2, a case Am Heart J Plus, 8, 100042.doi:10.1016/j.ahjo.2021.100042. https://www.ncbi.nlm.nih.gov/pubmed/34396358
- Kwan, Y. W., Chua, G. T., Chow, C. B., Tsao, S. S. L., To, K. K. W., Yuen, K.
Y., . . . Ip, P. (2021). mRNA COVID vaccine and myocarditis in adolescents. Hong Kong Med J, 27(5), 326-327. doi:10.12809/hkmj215120. https:// www.ncbi.nlm.nih.gov/pubmed/34393110
- Lee, , Chew, N. W. S., Ng, P., & Yeo, T. J. (2021). Reply to “Letter to the editor: Myocarditis should be considered in those with a troponin rise and unobstructed coronary arteries following PfizerBioNTech COVID-19 vaccination”. QJM. doi: 10.1093/qjmed/hcab232. https://www.ncbi.nlm.nih.gov/pubmed/34463770
- Levin, , Shimon, G., Fadlon-Derai, M., Gershovitz, L., Shovali, A., Sebbag, A., . . . Gordon, B. (2021). Myocarditis following COVID-19 vaccination – A case series. Vaccine, 39(42), 6195-6200. doi:10.1016/j.vaccine.2021.09.004. https:// www.ncbi.nlm.nih.gov/pubmed/34535317
- Li, M., Yuan, J., Lv, G., Brown, J., Jiang, X., & Lu, Z. K. (2021). Myocarditis and Pericarditis following COVID-19 Vaccination: Inequalities in Age and Vaccine J Pers Med, 11(11). doi:10.3390/jpm11111106. https://www.ncbi.nlm.nih.gov/pubmed/ 34834458
- Lim, , Kim, M. C., Kim, K. H., Jeong, I. S., Cho, Y. S., Choi, Y. D., & Lee, J. E.
(2021). Case Report: Acute Fulminant Myocarditis and Cardiogenic Shock After Messenger RNA CoronavirusDisease 2019 Vaccination Requiring Extracorporeal Cardiopulmonary Resuscitation. Front Cardiovasc Med, 8, 758996. doi:10.3389/ fcvm.2021.758996. https://www.ncbi.nlm.nih.gov/pubmed/34778411
- Luk, , Clarke, B., Dahdah, N., Ducharme, A., Krahn, A., McCrindle, B., . . . McDonald, M. (2021). Myocarditis and Pericarditis After COVID-19 mRNA Vaccination: Practical Considerations for Care Providers. Can J Cardiol, 37(10), 1629-1634. doi:10.1016/j.cjca.2021.08.001.https://www.ncbi.nlm.nih.gov/pubmed/
34375696
- Mevorach, , Anis, E., Cedar, N., Bromberg, M., Haas, E. J., Nadir, E., . . . Alroy- Preis, S. (2021). Myocarditis after BNT162b2 mRNA Vaccine against Covid-19 in Israel. N Engl J Med, 385(23), 2140-2149. doi:10.1056/NEJMoa2109730. https:// www.ncbi.nlm.nih.gov/pubmed/34614328
- Minocha, P. K., Better, D., Singh, R. K., & Hoque, T. (2021). Recurrence of Acute Myocarditis TemporallyAssociated with Receipt of the mRNA Coronavirus Disease 2019 (COVID-19) Vaccine in a Male J Pediatr, 238, 321-323. doi: 10.1016/j.jpeds.2021.06.035. https://www.ncbi.nlm.nih.gov/pubmed/34166671
- Montgomery, , Ryan, M., Engler, R., Hoffman, D., McClenathan, B., Collins, L., . . . Cooper, L. T., Jr.(2021). Myocarditis Following Immunization With mRNA COVID-19 Vaccines in Members of the US Military. JAMA Cardiol, 6(10), 1202-1206. doi: 10.1001/jamacardio.2021.2833. https://www.ncbi.nlm.nih.gov/pubmed/34185045
- Murakami, , Shinohara, M., Oka, Y., Wada, R., Noike, R., Ohara, H., . . . Ikeda, T. (2021). MyocarditisFollowing a COVID-19 Messenger RNA Vaccination: A Japanese Case Series. Intern Med. doi:10.2169/internalmedicine.8731-21. https:// www.ncbi.nlm.nih.gov/pubmed/34840235
- Nagasaka, , Koitabashi, N., Ishibashi, Y., Aihara, K., Takama, N., Ohyama, Y., . . . Kaneko, Y. (2021).Acute Myocarditis Associated with COVID-19 Vaccination: A Case Report. J Cardiol Cases. doi:10.1016/j.jccase.2021.11.006. https:// www.ncbi.nlm.nih.gov/pubmed/34876937
- Premature myocardial infarction or side effect of COVID-19 vaccine: https://ncbi.nlm.nih.gov/33824804/
- the culprit: https://pubmed.ncbi.nlm.nih.gov/34702550/
- A case of acute encephalopathy and non-ST-segment elevation myocardial infarction aftervaccination with mRNA-1273: possible adverse effect: https:// ncbi.nlm.nih.gov/34703815/
- Acute myocardial infarction within 24 hours after COVID-19 vaccination: is Kounis syndrome Clinical and histopathologic spectrum of delayed adverse skin reactions after COVID-19 vaccination: https://pubmed.ncbi.nlm.nih.gov/34292611/.
- Chouchana, , Blet, A., Al-Khalaf, M., Kafil, T. S., Nair, G., Robblee, J., . . . Liu, P. P. (2021). Features of Inflammatory Heart Reactions Following mRNA COVID-19 Vaccination at a Global Level. Clin Pharmacol Ther. doi:10.1002/cpt.2499. https:// www.ncbi.nlm.nih.gov/pubmed/34860360
- Three cases of acute venous thromboembolism in women after vaccination against COVID-19: https://sciencedirect.com/science/article/pii/S2213333X21003929
- Acute thrombosis of the coronary tree after vaccination against COVID-19: https://sciencedirect.com/science/article/abs/pii/S1936879821003988
- US case reports of cerebral venous sinus thrombosis with thrombocytopenia after vaccination withCOV2.S (against covid-19), March 2 to April 21, 2020: https:// pubmed.ncbi.nlm.nih.gov/33929487/
- Portal vein thrombosis associated with ChAdOx1 nCov-19 vaccine: https://thelancet.com/journals/langas/article/PIIS2468-1253(21)00197-7/
- Management of cerebral and splanchnic vein thrombosis associated with thrombocytopenia in subjects previously vaccinated with Vaxzevria (AstraZeneca): position statement of the Italian Society for the Study ofHemostasis and Thrombosis (SISET): https://pubmed.ncbi.nlm.nih.gov/33871350/
- Thrombosis with thrombocytopenia syndrome associated with COVID-19 vaccines: https://sciencedirect.com/science/article/abs/pii/S0735675721004381
- Covid-19 vaccine-induced thrombosis and thrombocytopenia: a commentary on an important and practical clinical dilemma: https://sciencedirect.com/science/ article/abs/pii/S0033062021000505
- Thrombosis with thrombocytopenia syndrome associated with COVID-19 viral vector vaccines: https://sciencedirect.com/science/article/abs/pii/S0953620521001904
- COVID-19 vaccine-induced immune-immune thrombotic thrombocytopenia: an emerging cause ofsplanchnic vein thrombosis: https://sciencedirect.com/ science/article/pii/S1665268121000557
- The roles of platelets in COVID-19-associated coagulopathy and vaccine-induced immune thrombotic immune thrombocytopenia (covid): https:// sciencedirect.com/science/article/pii/S1050173821000967
- Roots of autoimmunity of thrombotic events after COVID-19 vaccination: https://sciencedirect.com/science/article/abs/pii/S1568997221002160
- Thrombotic immune thrombocytopenia induced by SARS-CoV-2 vaccine: https://nejm.org/doi/full/10.1056/nejme2106315
- Thrombosis and thrombocytopenia after vaccination with ChAdOx1 nCoV-19: https://nejm.org/doi/full/10.1056/NEJMoa2104882? query=recirc_curatedRelated_article
- Thrombotic thrombocytopenia after vaccination with ChAdOx1 nCov-19: https://nejm.org/doi/full/10.1056/NEJMoa2104840? query=recirc_curatedRelated_article
- Post-mortem findings in vaccine-induced thrombotic thrombocytopenia (covid-19):https://haematologica.org/article/view/haematol.2021.279075
- Comparison of vaccine-induced thrombotic episodes between ChAdOx1 nCoV-19 and Ad26.COV.2.S vaccines: https://sciencedirect.com/science/article/abs/pii/ S0896841121000895
- Hypothesis behind the very rare cases of thrombosis with thrombocytopenia syndrome after SARS-CoV-2 vaccination: https://sciencedirect.com/science/ article/abs/pii/S0049384821003315
- Primary adrenal insufficiency associated with thrombotic immune thrombocytopenia induced by the Oxford-AstraZeneca ChAdOx1 nCoV-19 vaccine (VITT): https://sciencedirect.com/science/article/pii/S0953620521002363
- “Portal vein thrombosis occurring after the first dose of SARS-CoV-2 mRNA vaccine in a patient withantiphospholipid syndrome”: https://sciencedirect.com/science/ article/pii/S2666572721000389
- Early results of bivalirudin treatment for thrombotic thrombocytopenia and cerebral venous sinus thrombosis after vaccination with Ad26.COV2.S: https://sciencedirect.com/science/article/pii/S0196064421003425
- Mechanisms of immunothrombosis in vaccine-induced thrombotic thrombocytopenia (VITT) compared to natural SARS-CoV-2 infection: https://sciencedirect.com/ science/article/abs/pii/S0896841121000706
- Prothrombotic immune thrombocytopenia after COVID-19 vaccination: https://sciencedirect.com/science/article/pii/S0006497121009411
- Vaccine-induced thrombotic thrombocytopenia: the dark chapter of a success story: https://sciencedirect.com/science/article/pii/S2589936821000256
- Thrombosis after COVID-19 vaccination: possible link to ACE pathways: https://sciencedirect.com/science/article/pii/S0049384821004369
- Vaccine-induced thrombotic thrombocytopenia, a rare but severe case of friendly fire in the battle against the COVID-19 pandemic: What pathogenesis?: https://sciencedirect.com/science/article/pii/S0953620521002314
- Thrombocytopenia and intracranial venous sinus thrombosis after exposure to the “AstraZeneca COVID-19 vaccine”: https://pubmed.ncbi.nlm.nih.gov/33918932/
- Thrombosis with thrombocytopenia after messenger RNA vaccine -1273: https://ncbi.nlm.nih.gov/34181446/
- First dose of ChAdOx1 and BNT162b2 COVID-19 vaccines and thrombocytopenic, thromboembolic, and hemorrhagic events in Scotland: https://nature.com/ articles/s41591-021-01408-4
- PF4 immunoassays in vaccine-induced thrombotic thrombocytopenia: https://nejm.org/doi/full/10.1056/NEJMc2106383
- Antibody epitopes in vaccine-induced immune immune thrombotic thrombocytopenia: https://nature.com/articles/s41586-021-03744-4
- Thrombosis with thrombocytopenia syndrome associated with COVID-19 vaccines:. https://sciencedirect.com/science/article/abs/pii/S0735675721004381.
- Immune thrombosis and thrombocytopenia (VITT) associated with the COVID-19 vaccine: diagnostic andtherapeutic recommendations for a new syndrome: https:// ncbi.nlm.nih.gov/33987882/
- Laboratory testing for suspicion of COVID-19 vaccine-induced thrombotic (immune) thrombocytopenia: https://pubmed.ncbi.nlm.nih.gov/34138513/
- Intracerebral haemorrhage due to thrombosis with thrombocytopenia syndrome after COVID-19vaccination: the first fatal case in Korea: https://pubmed.ncbi.nlm.nih.gov/ 34402235/
- Risk of thrombocytopenia and thromboembolism after covid-19 vaccination and positive SARS-CoV-2 tests: self-controlled case series study: https:// ncbi.nlm.nih.gov/34446426/
- Vaccine-induced immune thrombotic thrombocytopenia and cerebral venous sinus thrombosis after covid-19 vaccination; a systematic review: https:// ncbi.nlm.nih.gov/34365148/.
- Primary adrenal insufficiency associated with thrombotic immune thrombocytopenia induced by Oxford-AstraZeneca ChAdOx1 nCoV-19 vaccine (VITT): https:// ncbi.nlm.nih.gov/34256983/
- Thromboaspiration infusion and fibrinolysis for portomesenteric thrombosis after administration ofAstraZeneca COVID-19 vaccine: https://pubmed.ncbi.nlm.nih.gov/ 34132839/
- 59-year-old woman with extensive deep venous thrombosis and pulmonary thromboembolism 7 daysafter a first dose of Pfizer-BioNTech BNT162b2 mRNA vaccine COVID-19: https://pubmed.ncbi.nlm.nih.gov/34117206/
- Thrombosis with thrombocytopenia syndrome (TTS) following AstraZeneca ChAdOx1 nCoV-19 (AZD1222) COVID-19 vaccination: risk-benefit analysis for persons <60 years in Australia: https://pubmed.ncbi.nlm.nih.gov/34272095/
- Comparison of vaccine-induced thrombotic events between ChAdOx1 nCoV-19 and Ad26.COV.2.S vaccines: https://pubmed.ncbi.nlm.nih.gov/34139631/.
- Bilateral superior ophthalmic vein thrombosis, ischemic stroke and immune thrombocytopenia after vaccination with ChAdOx1 nCoV-19: https:// ncbi.nlm.nih.gov/33864750/
- celiac artery and splenic artery thrombosis complicated by splenic infarction 7 days after the first dose of Oxford vaccine, causal relationship or coincidence: https:// ncbi.nlm.nih.gov/34261633/.
- Primary adrenal insufficiency associated with Oxford-AstraZeneca ChAdOx1 nCoV-19 (VITT) vaccine-induced immune thrombotic thrombocytopenia: https:// ncbi.nlm.nih.gov/34256983/
- Thrombosis with thrombocytopenia syndrome after COVID-19 immunization: https://ncbi.nlm.nih.gov/34236343/
- Thrombosis with thrombocytopenia syndrome associated with COVID-19 viral vector vaccines: https://pubmed.ncbi.nlm.nih.gov/34092488/
- Thromboaspiration infusion and fibrinolysis for portomesenteric thrombosis after administration of the AstraZeneca COVID-19 vaccine: https:// ncbi.nlm.nih.gov/34132839/.
- Atypical thrombosis associated with the vaccine VaxZevria® (AstraZeneca): data from the French network of regional pharmacovigilance centers: https:// ncbi.nlm.nih.gov/34083026/.
- Vaccine-induced thrombosis and thrombocytopenia with bilateral adrenal haemorrhage: https://pubmed.ncbi.nlm.nih.gov/34235757/.
- Palmar digital vein thrombosis after Oxford-AstraZeneca COVID-19 vaccination: https://pubmed.ncbi.nlm.nih.gov/34473841/.
- Cutaneous thrombosis associated with cutaneous necrosis following Oxford- AstraZeneca COVID-19vaccination: https://pubmed.ncbi.nlm.nih.gov/34189756/
- Thrombosis with thrombocytopenia after Messenger vaccine RNA-1273: https://ncbi.nlm.nih.gov/34181446/
- Coronavirus (COVID-19) Vaccine-induced immune thrombotic thrombocytopenia (VITT): https://pubmed.ncbi.nlm.nih.gov/34033367/
- Comparison of adverse drug reactions among four COVID-19 vaccines in Europe using the EudraVigilance database: Thrombosis in unusual sites: https:// ncbi.nlm.nih.gov/34375510/
- Immunoglobulin adjuvant for vaccine-induced immune thrombotic thrombocytopenia: https://pubmed.ncbi.nlm.nih.gov/34107198/
- Severe vaccine-induced thrombotic thrombocytopenia following vaccination with COVID-19: an autopsy case report and review of the literature: https:// ncbi.nlm.nih.gov/34355379/.
- Platelet activation and modulation in thrombosis with thrombocytopenia syndrome associated with theChAdO × 1 nCov-19 vaccine: https://pubmed.ncbi.nlm.nih.gov/ 34474550/
- Report of the International Cerebral Venous Thrombosis Consortium on cerebral venous thrombosis afterSARS-CoV-2 vaccination: https://pubmed.ncbi.nlm.nih.gov/ 34462996/
- Immune thrombocytopenia associated with the Pfizer-BioNTech COVID-19 mRNA vaccine BNT162b2: https://sciencedirect.com/science/article/pii/ S2214250921002018
- Secondary immune thrombocytopenia putatively attributable to COVID-19 vaccination: https://casereports.bmj.com/content/14/5/e242220.abstract.
- Immune thrombocytopenia following Pfizer-BioNTech BNT162b2 mRNA COVID-19 vaccine: https://pubmed.ncbi.nlm.nih.gov/34155844/
- Newly diagnosed idiopathic thrombocytopenia after COVID-19 vaccine administration: https://ncbi.nlm.nih.gov/pmc/articles/PMC8176657/.
- Idiopathic thrombocytopenic purpura and the Modern Covid-19 vaccine: https://annemergmed.com/article/S0196-0644(21)00122-0/fulltext.
- Thrombocytopenia after Pfizer and Moderna SARS vaccination – CoV -2: https://ncbi.nlm.nih.gov/pmc/articles/PMC8014568/.
- Immune thrombocytopenic purpura and acute liver injury after COVID-19 vaccination: https://casereports.bmj.com/content/14/7/e242678.
- Carotid artery immune thrombosis induced by adenovirus-vectored COVID-19 vaccine: case report: https://pubmed.ncbi.nlm.nih.gov/34312301/.
- The roles of platelets in COVID-19-associated coagulopathy and vaccine-induced immune-immune thrombotic thrombocytopenia: https://pubmed.ncbi.nlm.nih.gov/ 34455073/
- Cerebral venous sinus thrombosis negative for anti-PF4 antibody without thrombocytopenia after immunization with COVID-19 vaccine in a non-comorbid elderly Indian male treated with conventionalheparin-warfarin-based anticoagulation: https://pubmed.ncbi.nlm.nih.gov/34186376/
- Arterial events, venous thromboembolism, thrombocytopenia and bleeding after vaccination with Oxford-AstraZeneca ChAdOx1-S in Denmark and Norway: population-based cohort study: https://pubmed.ncbi.nlm.nih.gov/33952445/
- Procoagulant microparticles: a possible link between vaccine-induced immune thrombocytopenia (VITT) and cerebral sinus venous thrombosis: https:// ncbi.nlm.nih.gov/34129181/
- S. case reports of cerebral venous sinus thrombosis with thrombocytopenia after vaccination with Ad26.COV2.S, March 2-April 21, 2021: https:// pubmed.ncbi.nlm.nih.gov/33929487/.
- Malignant cerebral infarction after vaccination with ChAdOx1 nCov-19: a catastrophic variant of vaccine-induced immune-mediated thrombotic thrombocytopenia: https:// ncbi.nlm.nih.gov/34341358/
- Acute ischemic stroke revealing immune thrombotic thrombocytopenia induced by ChAdOx1 nCov-19 vaccine: impact on recanalization strategy: https:// ncbi.nlm.nih.gov/34175640/
- Vaccine-induced immune thrombotic immune thrombocytopenia (VITT): a new clinicopathologic entity with heterogeneous clinical presentations: https:// ncbi.nlm.nih.gov/34159588/.
- Imaging and hematologic findings in thrombosis and thrombocytopenia after vaccination with ChAdOx1nCoV-19 (AstraZeneca): https://pubmed.ncbi.nlm.nih.gov/ 34402666/
- Autoimmunity roots of thrombotic events after vaccination with COVID-19: https://ncbi.nlm.nih.gov/34508917/
- Cerebral venous sinus thrombosis after vaccination: the UK experience: https://ncbi.nlm.nih.gov/34370974/
- Cutaneous thrombosis associated with cutaneous necrosis following Oxford- AstraZeneca COVID-19vaccination: https://pubmed.ncbi.nlm.nih.gov/34189756/
- Myocardial infarction and azygos vein thrombosis after vaccination with ChAdOx1 nCoV-19 in a hemodialysis patient: https://pubmed.ncbi.nlm.nih.gov/34650896/
- Refractory vaccine-induced immune thrombotic thrombocytopenia (VITT) treated with delayed therapeutic plasma exchange (TPE): https://pubmed.ncbi.nlm.nih.gov/ 34672380/.
- Rare case of COVID-19 vaccine-associated intracranial hemorrhage with venous sinus thrombosis: https://pubmed.ncbi.nlm.nih.gov/34556531/.
- Delayed headache after COVID-19 vaccination: a warning sign for vaccine-induced cerebral venous thrombosis: https://pubmed.ncbi.nlm.nih.gov/34535076/.
- Clinical features of vaccine-induced thrombocytopenia and immune thrombosis: https://pubmed.ncbi.nlm.nih.gov/34379914/.
- Predictors of mortality in thrombotic thrombocytopenia after adenoviral COVID-19 vaccination: the FAPIC score: https://pubmed.ncbi.nlm.nih.gov/34545400/
- Ischemic stroke as a presenting feature of immune thrombotic thrombocytopenia induced by ChAdOx1-nCoV-19 vaccination: https://pubmed.ncbi.nlm.nih.gov/ 34035134/
- Endovascular treatment for vaccine-induced cerebral venous sinus thrombosis and thrombocytopenia after vaccination with ChAdOx1 nCoV-19: report of three cases: https://pubmed.ncbi.nlm.nih.gov/34782400/
- Possible triggers of thrombocytopenia and/or hemorrhage by BNT162b2 vaccine, Pfizer-BioNTech: https://pubmed.ncbi.nlm.nih.gov/34660652/.
- Multiple sites of arterial thrombosis in a 35-year-old patient after vaccination with ChAdOx1(AstraZeneca), which required emergency femoral and carotid surgical thrombectomy: https://pubmed.ncbi.nlm.nih.gov/34644642/
- Case series of vaccine-induced thrombotic thrombocytopenia in a London teaching hospital: https://pubmed.ncbi.nlm.nih.gov/34694650/
- Neuro-ophthalmic complications with thrombocytopenia and thrombosis induced by ChAdOx1 nCoV-19 vaccine: https://pubmed.ncbi.nlm.nih.gov/34726934/
- Thrombotic events after COVID-19 vaccination in over 50 years of age: results of a population-based study in Italy: https://pubmed.ncbi.nlm.nih.gov/34835237/
- Intracerebral hemorrhage associated with vaccine-induced thrombotic thrombocytopenia afterChAdOx1 nCOVID-19 vaccination in a pregnant woman: https://pubmed.ncbi.nlm.nih.gov/34261297/
- Age- and sex-specific incidence of cerebral venous sinus thrombosis associated with Ad26.COV2.S COVID-19 vaccination: https://pubmed.ncbi.nlm.nih.gov/34724036/.
- Genital necrosis with cutaneous thrombosis following vaccination with COVID-19 mRNA: https://pubmed.ncbi.nlm.nih.gov/34839563/
- Cerebral venous sinus thrombosis after mRNA-based COVID-19 vaccination: https://ncbi.nlm.nih.gov/34783932/.
- COVID-19 vaccine-induced immune thrombosis with thrombocytopenia thrombosis (VITT) and shades ofgray in thrombus formation: https://pubmed.ncbi.nlm.nih.gov/ 34624910/
- Acute ST-segment elevation myocardial infarction secondary to vaccine-induced immune thrombosis withthrombocytopenia (VITT): https://pubmed.ncbi.nlm.nih.gov/ 34580132/
- Thrombosis with thrombocytopenia syndrome (TTS) after vaccination with AstraZeneca ChAdOx1nCoV-19 (AZD1222) COVID-19: a risk-benefit analysis for persons <60% risk-benefit analysis for people <60 years in Australia: https:// ncbi.nlm.nih.gov/34272095/
- Characteristics and outcomes of patients with cerebral venous sinus thrombosis in thrombotic immune thrombocytopenia induced by SARS-CoV-2 vaccine: https://com/journals/jamaneurology/fullarticle/2784622
- Case study of thrombosis and thrombocytopenia syndrome after administration of the AstraZeneca COVID-19 vaccine: https://pubmed.ncbi.nlm.nih.gov/34781321/
- Thrombosis with Thrombocytopenia Syndrome Associated with COVID-19 Vaccines: https://pubmed.ncbi.nlm.nih.gov/34062319/
- Cerebral venous sinus thrombosis following vaccination with ChAdOx1: the first case of definite thrombosis with thrombocytopenia syndrome in India: https:// ncbi.nlm.nih.gov/34706921/
- COVID-19 vaccine-associated thrombosis with thrombocytopenia syndrome (TTS): systematic review andpost hoc analysis: https://pubmed.ncbi.nlm.nih.gov/34698582/
- Concerns for adverse effects of thrombocytopenia and thrombosis after adenovirus- vectored COVID-19 vaccination: https://pubmed.ncbi.nlm.nih.gov/34541935/
- Cerebral venous sinus thrombosis after COVID-19 vaccination: neurologic and radiologic management: https://pubmed.ncbi.nlm.nih.gov/34327553/.
- Case report: cerebral sinus vein thrombosis in two patients with AstraZeneca SARS- CoV-2 vaccine: https://pubmed.ncbi.nlm.nih.gov/34609603/
- Vaccine-induced immune thrombosis and thrombocytopenia syndrome after adenovirus-vectored severeacute respiratory syndrome coronavirus 2 vaccination: a new hypothesis on mechanisms and implications for future vaccine development: https://pubmed.ncbi.nlm.nih.gov/34664303/.
- Thrombosis in peripheral artery disease and thrombotic thrombocytopenia following adenoviral COVID-19 vaccination: https://pubmed.ncbi.nlm.nih.gov/34649281/
- Cerebral venous sinus thrombosis and thrombotic events after vector-based COVID-19 vaccines: systematic review and meta-analysis: https:// ncbi.nlm.nih.gov/34610990/.
- Thrombosis after COVID-19 vaccination: possible link to ACE pathways: https://ncbi.nlm.nih.gov/34479129/
- Major artery thrombosis and vaccination against ChAdOx1 nCov-19: https://ncbi.nlm.nih.gov/34839830/
- Understanding the risk of thrombosis with thrombocytopenia syndrome following Ad26.COV2.S vaccination: https://pubmed.ncbi.nlm.nih.gov/34595694/
- Comments on thrombosis after vaccination: spike protein leader sequence could be responsible for thrombosis and antibody-mediated thrombocytopenia: https:// ncbi.nlm.nih.gov/34788138
- Thrombosis in pre- and post-vaccination phase of COVID-19; https://ncbi.nlm.nih.gov/34650382/
- Vaccine-associated thrombocytopenia and thrombosis: venous endotheliopathy leading to combined venous micro-macrothrombosis: https:// ncbi.nlm.nih.gov/34833382/
- Thrombosis and thrombocytopenia syndrome causing isolated symptomatic carotid occlusion after COVID-19 Ad26.COV2.S vaccine (Janssen): https:// ncbi.nlm.nih.gov/34670287/
- An unusual presentation of acute deep vein thrombosis after Modern COVID-19 vaccine: case report: https://pubmed.ncbi.nlm.nih.gov/34790811/
- Immediate high-dose intravenous immunoglobulins followed by direct treatment with thrombin inhibitors is crucial for survival in vaccine-induced immune thrombotic thrombocytopenia Sars-Covid-19-vector adenoviral VITT with venous thrombosis of the cerebral sinus and portal vein: https://pubmed.ncbi.nlm.nih.gov/34023956/.
- Thrombosis formation after COVID-19 vaccination immunologic aspects: review article: https://pubmed.ncbi.nlm.nih.gov/34629931/
- Imaging and hematologic findings in thrombosis and thrombocytopenia after vaccination with ChAdOx1nCoV-19 (AstraZeneca): https://pubmed.ncbi.nlm.nih.gov/ 34402666/
- Cerebral venous sinus thrombosis, pulmonary embolism, and thrombocytopenia after COVID-19vaccination in a Taiwanese man: a case report and review of the literature: https://pubmed.ncbi.nlm.nih.gov/34630307/
- Fatal cerebral venous sinus thrombosis after COVID-19 vaccination: https://ncbi.nlm.nih.gov/33983464/
- Autoimmune roots of thrombotic events after COVID-19 vaccination: https://ncbi.nlm.nih.gov/34508917/.
- New portal vein thrombosis in cirrhosis: is thrombophilia exacerbated by vaccine or COVID-19: https://jcehepatology.com/article/S0973-6883(21)00545-4/fulltext.
- Cerebral venous sinus thrombosis after vaccination with COVID-19 mRNA of BNT162b2: https://pubmed.ncbi.nlm.nih.gov/34796065/.
- A case of unusual mild clinical presentation of COVID-19 vaccine-induced immune thrombotic thrombocytopenia with splanchnic vein thrombosis: https:// ncbi.nlm.nih.gov/34843991/
- Cerebral venous sinus thrombosis following vaccination with Pfizer-BioNTech COVID-19 (BNT162b2): https://pubmed.ncbi.nlm.nih.gov/34595867/
- A case of idiopathic thrombocytopenic purpura after a booster dose of COVID-19 BNT162b2 vaccine(Pfizer-Biontech): https://pubmed.ncbi.nlm.nih.gov/34820240/
- Vaccine-induced immune thrombotic immune thrombocytopenia (VITT): targeting pathologic mechanisms with Bruton’s tyrosine kinase inhibitors: https:// ncbi.nlm.nih.gov/33851389/
- Thromboembolic events in younger females exposed to Pfizer-BioNTech or Moderna COVID-19 vaccines: https://pubmed.ncbi.nlm.nih.gov/34264151/
- Thrombosis after adenovirus-vectored COVID-19 vaccination: a concern for underlying disease: https://pubmed.ncbi.nlm.nih.gov/34755555/
- Unusual site of deep vein thrombosis after vaccination against coronavirus mRNA-2019 coronavirusdisease (COVID-19): https://pubmed.ncbi.nlm.nih.gov/ 34840204/
- Deep venous thrombosis after vaccination with COV2.S in adult males: https://pubmed.ncbi.nlm.nih.gov/34659839/.
- Clinical and biological features of cerebral venous sinus thrombosis after vaccination with ChAdOx1 nCov-19; https://jnnp.bmj.com/content/early/2021/09/29/
jnnp-2021-327340.
- COV2-S vaccination may reveal hereditary thrombophilia: massive cerebral venous sinus thrombosis in a young man with normal platelet count: https:// pubmed.ncbi.nlm.nih.gov/34632750/
- Post-mortem findings in vaccine-induced thrombotic thrombocytopenia: https://org/article/view/haematol.2021.279075
- COVID-19 vaccine-induced thrombosis: https://pubmed.ncbi.nlm.nih.gov/34802488/.
- Inflammation and platelet activation after COVID-19 vaccines: possible mechanisms behind vaccine-induced immune thrombocytopenia and thrombosis: https:// ncbi.nlm.nih.gov/34887867/.
- Anaphylactoid reaction and coronary thrombosis related to COVID-19 mRNA vaccine: https://pubmed.ncbi.nlm.nih.gov/34863404/.
- Occurrence of splenic infarction due to arterial thrombosis after vaccination with COVID-19: https://pubmed.ncbi.nlm.nih.gov/34876440/
- Deep venous thrombosis more than two weeks after COVID-19 vaccination: https://ncbi.nlm.nih.gov/33928773/
- Information on ChAdOx1 nCoV-19 vaccine-induced immune-mediated thrombotic thrombocytopenia: https://pubmed.ncbi.nlm.nih.gov/34587242/
- Management of a patient with a rare congenital limb malformation syndrome after SARS-CoV-2 vaccine-induced thrombosis and thrombocytopenia (VITT): https:// ncbi.nlm.nih.gov/34097311/
- Thrombocytopenia and splanchnic thrombosis after vaccination with COV2.S successfully treated with transjugular intrahepatic intrahepatic portosystemic shunt and thrombectomy: https://onlinelibrary.wiley.com/doi/10.1002/ajh.26258
- Chang, C., & Hawley, H. B. (2021). Vaccine-Associated Thrombocytopenia and Thrombosis: Venous Endotheliopathy Leading to Venous Combined Micro- Macrothrombosis. Medicina (Kaunas), 57(11). doi:10.3390/medicina57111163. https://www.ncbi.nlm.nih.gov/pubmed/34833382
- Potential risk of thrombotic events after COVID-19 vaccination with Oxford- AstraZeneca inwomen receiving estrogen: https://pubmed.ncbi.nlm.nih.gov/ 34734086/
- Thrombotic adverse events reported for Moderna, Pfizer, and Oxford-AstraZeneca COVID-19 vaccines: comparison of occurrence and clinical outcomes in the EudraVigilance database: https://pubmed.ncbi.nlm.nih.gov/34835256/
- Predicted and observed incidence of thromboembolic events among Koreans vaccinated with theChAdOx1 nCoV-19 vaccine: https://pubmed.ncbi.nlm.nih.gov/ 34254476/
- Three cases of acute venous thromboembolism in women after coronavirus 2019 vaccination: https://pubmed.ncbi.nlm.nih.gov/34352418/
- Shazley, , & Alshazley, M. (2021). A COVID-Positive 52-Year-Old Man Presented With Venous Thromboembolism and Disseminated Intravascular Coagulation Following Johnson & Johnson Vaccination:A Case-Study. Cureus, 13(7), e16383. doi:10.7759/cureus.16383. https://www.ncbi.nlm.nih.gov/pubmed/34408937
- Vaccine-induced immune thrombotic thrombocytopenia with disseminated intravascular coagulationand death after ChAdOx1 nCoV-19 vaccination: https:// ncbi.nlm.nih.gov/34171649/
- US case reports of cerebral venous sinus thrombosis with thrombocytopenia after vaccination withCOV2.S (against covid-19), March 2 to April 21, 2020: https:// pubmed.ncbi.nlm.nih.gov/33929487/
- Management of cerebral and splanchnic vein thrombosis associated with thrombocytopenia in subjects previously vaccinated with Vaxzevria (AstraZeneca): position statement of the Italian Society for the Study ofHemostasis and Thrombosis (SISET): https://pubmed.ncbi.nlm.nih.gov/33871350/
- Vaccine-induced immune thrombotic thrombocytopenia and cerebral venous sinus thrombosis after vaccination with COVID-19; a systematic review: https://sciencedirect.com/science/article/pii/S0022510X21003014
- Thrombosis with thrombocytopenia syndrome associated with COVID-19 vaccines: https://sciencedirect.com/science/article/abs/pii/S0735675721004381
- Covid-19 vaccine-induced thrombosis and thrombocytopenia: a commentary on an important and practical clinical dilemma: https://sciencedirect.com/science/ article/abs/pii/S0033062021000505
- Thrombosis with thrombocytopenia syndrome associated with COVID-19 viral vector vaccines: https://sciencedirect.com/science/article/abs/pii/S0953620521001904
- COVID-19 vaccine-induced immune-immune thrombotic thrombocytopenia: an emerging cause ofsplanchnic vein thrombosis: https://sciencedirect.com/ science/article/pii/S1665268121000557
- The roles of platelets in COVID-19-associated coagulopathy and vaccine-induced immune thrombotic immune thrombocytopenia (covid): https:// sciencedirect.com/science/article/pii/S1050173821000967
- Thrombotic immune thrombocytopenia induced by SARS-CoV-2 vaccine: https://nejm.org/doi/full/10.1056/nejme2106315
- Thrombosis and thrombocytopenia after vaccination with ChAdOx1 nCoV-19: https://nejm.org/doi/full/10.1056/NEJMoa2104882? query=recirc_curatedRelated_article
- Post-mortem findings in vaccine-induced thrombotic thrombocytopenia (covid-19):https://haematologica.org/article/view/haematol.2021.279075
- Thrombocytopenia, including immune thrombocytopenia after receiving COVID-19 mRNA vaccinesreported to the Vaccine Adverse Event Reporting System (VAERS):https://sciencedirect.com/science/article/pii/S0264410X21005247
- Hypothesis behind the very rare cases of thrombosis with thrombocytopenia syndrome after SARS-CoV-2 vaccination: https://sciencedirect.com/science/ article/abs/pii/S0049384821003315
- Primary adrenal insufficiency associated with thrombotic immune thrombocytopenia induced by the Oxford-AstraZeneca ChAdOx1 nCoV-19 vaccine (VITT): https://sciencedirect.com/science/article/pii/S0953620521002363
- Early results of bivalirudin treatment for thrombotic thrombocytopenia and cerebral venous sinus thrombosis after vaccination with Ad26.COV2.S: https://sciencedirect.com/science/article/pii/S0196064421003425
- Mechanisms of immunothrombosis in vaccine-induced thrombotic thrombocytopenia (VITT) compared to natural SARS-CoV-2 infection: https://sciencedirect.com/ science/article/abs/pii/S0896841121000706
- Vaccine-induced thrombotic thrombocytopenia: the dark chapter of a success story: https://sciencedirect.com/science/article/pii/S2589936821000256
- Cerebral venous sinus thrombosis and thrombocytopenia after COVID-19 vaccination: report of two cases in the United Kingdom: https://sciencedirect.com/science/article/abs/pii/S088915912100163X
- Immune thrombocytopenic purpura after vaccination with COVID-19 vaccine (ChAdOx1 nCov-19): https://sciencedirect.com/science/article/abs/pii/ S0006497121013963.
- Vaccine-induced thrombotic thrombocytopenia, a rare but severe case of friendly fire in the battle against the COVID-19 pandemic: What pathogenesis?: https://sciencedirect.com/science/article/pii/S0953620521002314
- Thrombocytopenia and intracranial venous sinus thrombosis after exposure to the “AstraZeneca COVID-19 vaccine”: https://pubmed.ncbi.nlm.nih.gov/33918932/
- Thrombocytopenia following Pfizer and Moderna SARS-CoV-2 vaccination: https://ncbi.nlm.nih.gov/33606296/
- Severe and refractory immune thrombocytopenia occurring after SARS-CoV-2 vaccination: https://pubmed.ncbi.nlm.nih.gov/33854395/
- Purpuric rash and thrombocytopenia after mRNA-1273 (Modern) COVID-19 vaccine: https://ncbi.nlm.nih.gov/pmc/articles/PMC7996471/
- Thrombosis with thrombocytopenia after messenger RNA vaccine -1273: https://ncbi.nlm.nih.gov/34181446/
- First dose of ChAdOx1 and BNT162b2 COVID-19 vaccines and thrombocytopenic, thromboembolic, and hemorrhagic events in Scotland: https://nature.com/ articles/s41591-021-01408-4
- Exacerbation of immune thrombocytopenia after COVID-19 vaccination: https://ncbi.nlm.nih.gov/34075578/
- PF4 immunoassays in vaccine-induced thrombotic thrombocytopenia: https://nejm.org/doi/full/10.1056/NEJMc2106383
- Antibody epitopes in vaccine-induced immune immune thrombotic thrombocytopenia: https://nature.com/articles/s41586-021-03744-4
- Thrombosis with thrombocytopenia syndrome associated with COVID-19 vaccines:. https://sciencedirect.com/science/article/abs/pii/S0735675721004381.
- Immune thrombocytopenia associated with Pfizer-BioNTech’s COVID-19 BNT162b2 mRNA vaccine: https://sciencedirect.com/science/article/pii/ S2214250921002018.
- Immune thrombosis and thrombocytopenia (VITT) associated with the COVID-19 vaccine: diagnostic andtherapeutic recommendations for a new syndrome: https:// ncbi.nlm.nih.gov/33987882/
- Laboratory testing for suspicion of COVID-19 vaccine-induced thrombotic (immune) thrombocytopenia: https://pubmed.ncbi.nlm.nih.gov/34138513/
- Intracerebral haemorrhage due to thrombosis with thrombocytopenia syndrome after COVID-19vaccination: the first fatal case in Korea: https://pubmed.ncbi.nlm.nih.gov/ 34402235/
- Risk of thrombocytopenia and thromboembolism after covid-19 vaccination and positive SARS-CoV-2 tests: self-controlled case series study: https:// ncbi.nlm.nih.gov/34446426/
- Vaccine-induced immune thrombotic thrombocytopenia and cerebral venous sinus thrombosis after covid-19 vaccination; a systematic review: https:// ncbi.nlm.nih.gov/34365148/.
- Primary adrenal insufficiency associated with thrombotic immune thrombocytopenia induced by Oxford-AstraZeneca ChAdOx1 nCoV-19 vaccine (VITT): https:// ncbi.nlm.nih.gov/34256983/
- Cerebral venous thrombosis and vaccine-induced a. Oxford- AstraZeneca COVID-19: a missed opportunity for a rapid return on experience: https://pubmed.ncbi.nlm.nih.gov/34033927/
- Immune thrombocytopenia in a 22-year-old post Covid-19 vaccine: https://ncbi.nlm.nih.gov/33476455/
- Secondary immune thrombocytopenia (ITP) associated with ChAdOx1 Covid-19 vaccine: case report: https://pubmed.ncbi.nlm.nih.gov/34377889/
- Thrombosis with thrombocytopenia syndrome (TTS) following AstraZeneca ChAdOx1 nCoV-19 (AZD1222) COVID-19 vaccination: risk-benefit analysis for persons <60 years in Australia: https://pubmed.ncbi.nlm.nih.gov/34272095/
- Bilateral superior ophthalmic vein thrombosis, ischemic stroke and immune thrombocytopenia after vaccination with ChAdOx1 nCoV-19: https:// ncbi.nlm.nih.gov/33864750/
- Risk of thrombocytopenia and thromboembolism after covid-19 vaccination and positive SARS-CoV-2 tests: self-controlled case series study: https:// ncbi.nlm.nih.gov/34446426/
- First dose of ChAdOx1 and BNT162b2 COVID-19 vaccines and thrombocytopenic, thromboembolic and hemorrhagic events in Scotland: https:// ncbi.nlm.nih.gov/34108714/
- Thrombocytopenia after COVID-19 vaccination: https://pubmed.ncbi.nlm.nih.gov/ 34332437/.
- A case of multiple thrombocytopenia and thrombosis following vaccination with ChAdOx1 nCoV-19 against SARS-CoV-2: https://pubmed.ncbi.nlm.nih.gov/ 34137813/
- Vaccine-induced thrombotic thrombocytopenia: the elusive link between thrombosis and adenovirus-based SARS-CoV-2 vaccines: https://pubmed.ncbi.nlm.nih.gov/ 34191218/
- Acute ischemic stroke revealing immune thrombotic thrombocytopenia induced by ChAdOx1 nCov-19 vaccine: impact on recanalization strategy: https:// ncbi.nlm.nih.gov/34175640/
- Procoagulant antibody-mediated procoagulant platelets in immune thrombotic thrombocytopenia associated with SARS-CoV-2 vaccination: https:// ncbi.nlm.nih.gov/34011137/.
- Thrombotic thrombocytopenia after vaccination with COVID-19: in search of the underlying mechanism: https://pubmed.ncbi.nlm.nih.gov/34071883/
- Thrombosis and SARS-CoV-2 vaccines: vaccine-induced immune thrombotic thrombocytopenia: https://pubmed.ncbi.nlm.nih.gov/34237213/.
- Acquired thrombotic thrombocytopenic thrombocytopenic purpura: a rare disease associated with theBNT162b2 vaccine: https://pubmed.ncbi.nlm.nih.gov/34105247/.
- Immune complexes, innate immunity and NETosis in ChAdOx1 vaccine-induced thrombocytopenia: https://pubmed.ncbi.nlm.nih.gov/34405870/.
- Immune-mediated thrombocytopenic purpura after Pfizer-BioNTech COVID-19 vaccine in an elderly woman: https://pubmed.ncbi.nlm.nih.gov/34513446/
- Immune-mediated thrombocytopenia associated with COV2.S vaccine (Janssen; Johnson &Johnson): https://pubmed.ncbi.nlm.nih.gov/34469919/.
- Transient thrombocytopenia with glycoprotein-specific platelet autoantibodies after vaccination with Ad26.COV2.S: case report: https://pubmed.ncbi.nlm.nih.gov/ 34516272/.
- COVID-19 vaccine, immune thrombotic thrombocytopenia, jaundice, hyperviscosity: concern in cases withunderlying hepatic problems: https://pubmed.ncbi.nlm.nih.gov/ 34509271/.
- Immune thrombocytopenia after vaccination during the COVID-19 pandemic: https://ncbi.nlm.nih.gov/34435486/
- Vaccine-induced thrombocytopenia with severe headache: https://ncbi.nlm.nih.gov/34525282/
- Vaccine-induced immune thrombotic thrombocytopenia causing a severe form of cerebral venous thrombosis with a high mortality rate: a case series: https:// ncbi.nlm.nih.gov/34393988/.
- Adenovirus interactions with platelets and coagulation and vaccine-associated autoimmune thrombocytopenia thrombosis syndrome: https:// ncbi.nlm.nih.gov/34407607/.
- Australian and New Zealand approach to the diagnosis and treatment of vaccine- induced immune thrombosis and immune thrombocytopenia: https:// ncbi.nlm.nih.gov/34490632/
- An observational study to identify the prevalence of thrombocytopenia and anti-PF4 / polyanion antibodies in Norwegian health care workers after COVID-19 vaccination: https://pubmed.ncbi.nlm.nih.gov/33909350/
- A rare case of thrombosis and thrombocytopenia of the superior ophthalmic vein after ChAdOx1 nCoV-19 vaccination against SARS-CoV-2: https:// ncbi.nlm.nih.gov/34276917/
- Thrombosis and severe acute respiratory syndrome Coronavirus 2 vaccines: vaccine-induced immune thrombotic thrombocytopenia: https:// ncbi.nlm.nih.gov/34237213/.
- Renal vein thrombosis and pulmonary embolism secondary to vaccine-induced thrombotic immunethrombocytopenia (VITT): https://pubmed.ncbi.nlm.nih.gov/ 34268278/.
- Limb ischemia and pulmonary artery thrombosis after ChAdOx1 nCoV-19 vaccine (Oxford-AstraZeneca): a case of vaccine-induced immune thrombotic thrombocytopenia: https://pubmed.ncbi.nlm.nih.gov/33990339/.
- A case of vaccine-induced immune-immune thrombotic thrombocytopenia with massive arteriovenousthrombosis: https://pubmed.ncbi.nlm.nih.gov/34059191/
- Thrombocytopenia in an adolescent with sickle cell anemia after COVID-19 vaccination: https://pubmed.ncbi.nlm.nih.gov/34331506/
- Vaccine-induced thrombocytopenia with severe headache: https://ncbi.nlm.nih.gov/34525282/
- ChAdOx1 interacts with CAR and PF4 with implications for thrombosis with thrombocytopenia syndrome:https://science.org/doi/10.1126/sciadv.abl8213
- Lethal vaccine-induced immune thrombotic immune thrombocytopenia (VITT) following announcementCOV2.S: first documented case outside the U.S.: https:// pubmed.ncbi.nlm.nih.gov/34626338/
- A prothrombotic thrombocytopenic disorder resembling heparin-induced thrombocytopenia aftercoronavirus-19 vaccination: https://europepmc.org/article/ PPR/PPR304469 435.
- VITT (vaccine-induced immune thrombotic thrombocytopenia) after vaccination with ChAdOx1 nCoV-19: https://pubmed.ncbi.nlm.nih.gov/34731555/
- Vaccine-induced immune thrombotic thrombocytopenia (VITT): a new clinicopathologic entitywith heterogeneous clinical presentations: https:// ncbi.nlm.nih.gov/34159588/
- Treatment of acute ischemic stroke associated with ChAdOx1 nCoV-19 vaccine- induced immunethrombotic thrombocytopenia: https://pubmed.ncbi.nlm.nih.gov/ 34461442/
- Cerebral venous sinus thrombosis after vaccination: the UK experience: https://ncbi.nlm.nih.gov/34370974/
- Cerebral venous vein/venous sinus thrombosis with thrombocytopenia syndrome after COVID-19 vaccination: https://pubmed.ncbi.nlm.nih.gov/34373413/
- Portal vein thrombosis due to vaccine-induced immune thrombotic immune thrombocytopenia (VITT)after Covid vaccination with ChAdOx1 nCoV-19: https:// ncbi.nlm.nih.gov/34598301/
- Spontaneous HIT syndrome: knee replacement, infection and parallels with vaccine- induced immune thrombotic thrombocytopenia: https://pubmed.ncbi.nlm.nih.gov/ 34144250/
- Thrombocytopenia with acute ischemic stroke and hemorrhage in a patient recently vaccinated with an adenoviral vector-based COVID-19 vaccine:. https:// ncbi.nlm.nih.gov/33877737/
- ChAdOx1 nCoV-19 vaccine-associated thrombocytopenia: three cases of immune thrombocytopenia after107,720 doses of ChAdOx1 vaccination in Thailand: https:// ncbi.nlm.nih.gov/34483267/.
- Pulmonary embolism, transient ischemic attack, and thrombocytopenia after Johnson & Johnson COVID-19 vaccine: https://pubmed.ncbi.nlm.nih.gov/34261635/
- Neurosurgical considerations with respect to decompressive craniectomy for intracerebral hemorrhage after SARS-CoV-2 vaccination in vaccine-induced thrombotic thrombocytopenia-VITT: https://pubmed.ncbi.nlm.nih.gov/34202817/
- Secondary thrombocytopenia after SARS-CoV-2 vaccination: case report of haemorrhage and hematoma after minor oral surgery: https:// ncbi.nlm.nih.gov/34314875/.
- Venous thromboembolism and mild thrombocytopenia after vaccination with ChAdOx1 nCoV-19: https://pubmed.ncbi.nlm.nih.gov/34384129/
- Fatal exacerbation of ChadOx1-nCoV-19-induced thrombotic thrombocytopenia syndrome after successful initial therapy with intravenous immunoglobulins: a rationale for monitoring immunoglobulin Glevels: https://pubmed.ncbi.nlm.nih.gov/ 34382387/
- A rare case of COVID-19 vaccine-induced thrombotic thrombocytopenia (VITT) affecting thevenosplanchnic and pulmonary arterial circulation from a UK district general hospital: https://pubmed.ncbi.nlm.nih.gov/34535492/
- COVID-19 vaccine-induced thrombotic thrombocytopenia: a case series: https://ncbi.nlm.nih.gov/34527501/
- Immune thrombocytopenia after immunization with Vaxzevria ChadOx1-S vaccine (AstraZeneca), Victoria, Australia: https://pubmed.ncbi.nlm.nih.gov/34756770/
- Case report of immune thrombocytopenia after vaccination with ChAdOx1 nCoV-19: https://pubmed.ncbi.nlm.nih.gov/34751013/.
- Thrombocytopenia with acute ischemic stroke and hemorrhage in a patient recently vaccinated with an adenoviral vector-based COVID-19 vaccine: https:// ncbi.nlm.nih.gov/33877737/
- Intracerebral hemorrhage and thrombocytopenia after AstraZeneca COVID-19 vaccine: clinical and diagnostic challenges of vaccine-induced thrombotic thrombocytopenia: https://pubmed.ncbi.nlm.nih.gov/34646685/
- Thrombocytopenia, including immune thrombocytopenia after receiving COVID-19 mRNA vaccinesreported to the Vaccine Adverse Event Reporting System (VAERS):https://pubmed.ncbi.nlm.nih.gov/34006408/
- Newly diagnosed immune thrombocytopenia in a pregnant patient after coronavirus disease 2019 vaccination: https://pubmed.ncbi.nlm.nih.gov/34420249/
- Thrombocytopenia in an adolescent with sickle cell anemia after COVID-19 vaccination: https://pubmed.ncbi.nlm.nih.gov/34331506/
- Abdominal pain and bilateral adrenal hemorrhage from immune thrombotic thrombocytopenia induced byCOVID-19 vaccine: https://pubmed.ncbi.nlm.nih.gov/ 34546343/
- Venous thromboembolism and mild thrombocytopenia after ChAdOx1 nCoV-19 vaccination: https://pubmed.ncbi.nlm.nih.gov/34384129/.
- Severe immune thrombocytopenia following COVID-19 vaccination: report of four cases and review of the literature: https://pubmed.ncbi.nlm.nih.gov/34653943/.
- Relapse of immune thrombocytopenia after COVID-19 vaccination: https://ncbi.nlm.nih.gov/34591991/
- Images of immune thrombotic thrombocytopenia induced by Oxford / AstraZeneca® COVID-19 vaccine: https://pubmed.ncbi.nlm.nih.gov/33962903/.
- Adenovirus interactions with platelets and coagulation and vaccine-induced immune thromboticthrombocytopenia syndrome: https://pubmed.ncbi.nlm.nih.gov/34407607/
- Complicated case report of long-term vaccine-induced thrombotic immune thrombocytopenia A: https://pubmed.ncbi.nlm.nih.gov/34835275/.
- Prevalence of thrombocytopenia, anti-platelet factor 4 antibodies, and elevated D- dimer in Thais after vaccination with ChAdOx1 nCoV-19: https:// ncbi.nlm.nih.gov/34568726/
- Bilateral thalamic stroke: a case of COVID-19 (VITT) vaccine-induced immune thromboticthrombocytopenia or a coincidence due to underlying risk factors: https://ncbi.nlm.nih.gov/34820232/.
- Successful treatment of vaccine-induced immune thrombotic thrombocytopenia in a 26-year-old female patient: https://pubmed.ncbi.nlm.nih.gov/34614491/
- Case report: vaccine-induced immune thrombotic thrombocytopenia in a patient with pancreatic cancer after vaccination with messenger RNA-1273: https:// ncbi.nlm.nih.gov/34790684/
- Vaccine-induced thrombotic thrombocytopenia after Ad26.COV2.S vaccination in a man presenting asacute venous thromboembolism: https://pubmed.ncbi.nlm.nih.gov/ 34096082/
- Helms, M., Ansteatt, K. T., Roberts, J. C., Kamatam, S., Foong, K. S., Labayog, J. S., & Tarantino, M. D. (2021). Severe, Refractory Immune Thrombocytopenia Occurring After SARS-CoV-2 Vaccine. J Blood Med, 12, 221-224. doi:10.2147/ JBM.S307047. https://www.ncbi.nlm.nih.gov/pubmed/33854395
- Hippisley-Cox, , Patone, M., Mei, X. W., Saatci, D., Dixon, S., Khunti, K., . . .
Coupland, C. A. C. (2021). Risk of thrombocytopenia and thromboembolism after covid-19 vaccinationand SARS-CoV-2 positive testing: self-controlled case series study. BMJ, 374, n1931. doi:10.1136/bmj.n1931. https://www.ncbi.nlm.nih.gov/ pubmed/34446426
- Lee, J., Cines, D. B., Gernsheimer, T., Kessler, C., Michel, M., Tarantino, M.
D., . . . Bussel, J. B. (2021). Thrombocytopenia following Pfizer and Moderna SARS- CoV-2 vaccination. Am J Hematol, 96(5), 534-537. doi:10.1002/ajh.26132. https:// www.ncbi.nlm.nih.gov/pubmed/33606296
- Welsh, J., Baumblatt, J., Chege, W., Goud, R., & Nair, N. (2021).
Thrombocytopenia including immune thrombocytopenia after receipt of mRNA COVID-19 vaccines reported to the Vaccine Adverse Event Reporting System (VAERS). Vaccine, 39(25), 3329-3332.doi:10.1016/j.vaccine.2021.04.054. https:// www.ncbi.nlm.nih.gov/pubmed/34006408
- Relapse of immune thrombocytopenia after covid-19 vaccination in young male patient: https://pubmed.ncbi.nlm.nih.gov/34804803/.
- Peduncular, symptomatic cavernous bleeding after immune thrombocytopenia- induced SARS-CoV-2vaccination: https://pubmed.ncbi.nlm.nih.gov/34549178/.
- Cerebral venous sinus thrombosis in the U.S. population after SARS-CoV-2 vaccination with adenovirusand after COVID-19: https://sciencedirect.com/ science/article/pii/S0735109721051949
- Cerebral venous sinus thrombosis negative for anti-PF4 antibody without thrombocytopenia after immunization with COVID-19 vaccine in a non-comorbid elderly Indian male treated with conventionalheparin-warfarin based anticoagulation:https://sciencedirect.com/science/article/pii/S1871402121002046
- Cerebral venous thrombosis after BNT162b2 mRNA SARS-CoV-2 vaccine: https://sciencedirect.com/science/article/abs/pii/S1052305721003098
- Cerebral venous sinus thrombosis after vaccination: the United Kingdom experience: https://thelancet.com/journals/lancet/article/PIIS0140-6736(21)01788-8/fulltext
- US case reports of cerebral venous sinus thrombosis with thrombocytopenia after vaccination withCOV2.S (against covid-19), March 2 to April 21, 2020: https:// pubmed.ncbi.nlm.nih.gov/33929487/
- Management of cerebral and splanchnic vein thrombosis associated with thrombocytopenia in subjects previously vaccinated with Vaxzevria (AstraZeneca): position statement of the Italian Society for the Study ofHemostasis and Thrombosis (SISET): https://pubmed.ncbi.nlm.nih.gov/33871350/
- Vaccine-induced immune thrombotic thrombocytopenia and cerebral venous sinus thrombosis after vaccination with COVID-19; a systematic review: https://sciencedirect.com/science/article/pii/S0022510X21003014
- Early results of bivalirudin treatment for thrombotic thrombocytopenia and cerebral venous sinus thrombosis after vaccination with Ad26.COV2.S: https://sciencedirect.com/science/article/pii/S0196064421003425
- A rare case of a middle-aged Asian male with cerebral venous thrombosis after AstraZeneca COVID-19vaccination: https://sciencedirect.com/science/article/ pii/S0735675721005714
- Cerebral venous sinus thrombosis and thrombocytopenia after COVID-19 vaccination: report of two cases in the United Kingdom: https://sciencedirect.com/science/article/abs/pii/S088915912100163X
- Diagnostic-therapeutic recommendations of the ad-hoc FACME expert working group on the management ofcerebral venous thrombosis related to COVID-19 vaccination: https://sciencedirect.com/science/article/pii/S0213485321000839
- COVID-19 vaccination: information on the occurrence of arterial and venous thrombosis using data fromVigiBase: https://pubmed.ncbi.nlm.nih.gov/33863748/
- Cerebral venous thrombosis associated with the covid-19 vaccine in Germany: https://onlinelibrary.wiley.com/doi/10.1002/ana.26172
- Cerebral venous thrombosis following BNT162b2 mRNA vaccination of BNT162b2 against SARS-CoV-2: a black swan event: https://pubmed.ncbi.nlm.nih.gov/ 34133027/
- The importance of recognizing cerebral venous thrombosis following anti-COVID-19 vaccination: https://pubmed.ncbi.nlm.nih.gov/34001390/
- Cerebral venous sinus thrombosis negative for anti-PF4 antibody without thrombocytopenia afterimmunization with COVID-19 vaccine in an elderly, non- comorbid Indian male treated with conventional heparin-warfarin-based anticoagulation:. https://sciencedirect.com/science/article/pii/S1871402121002046.
- Vaccine-induced immune thrombotic thrombocytopenia and cerebral venous sinus thrombosis after covid-19 vaccination; a systematic review: https:// ncbi.nlm.nih.gov/34365148/.
- A rare case of cerebral venous thrombosis and disseminated intravascular coagulation temporallyassociated with administration of COVID-19 vaccine: https:// ncbi.nlm.nih.gov/33917902/
- Acute cerebral venous thrombosis and pulmonary artery embolism associated with the COVID-19 vaccine: https://pubmed.ncbi.nlm.nih.gov/34247246/.
- Cerebral venous thrombosis and vaccine-induced a. Oxford- AstraZeneca COVID-19: a missed opportunity for a rapid return on experience: https://pubmed.ncbi.nlm.nih.gov/34033927/
- Diagnosis and treatment of cerebral venous sinus thrombosis with vaccine-induced immune-immune thrombotic thrombocytopenia: https://pubmed.ncbi.nlm.nih.gov/ 33914590/
- Venous sinus thrombosis after vaccination with ChAdOx1 nCov-19: https://ncbi.nlm.nih.gov/34420802/
- Cerebral venous sinus thrombosis following vaccination against SARS-CoV-2: an analysis of cases reported to the European Medicines Agency: https:// ncbi.nlm.nih.gov/34293217/
- Arterial events, venous thromboembolism, thrombocytopenia and bleeding after vaccination with Oxford-AstraZeneca ChAdOx1-S in Denmark and Norway: population-based cohort study: https://pubmed.ncbi.nlm.nih.gov/33952445/
- Cerebral venous thrombosis associated with COVID-19 vaccine in Germany: https://ncbi.nlm.nih.gov/34288044/
- Malignant cerebral infarction after vaccination with ChAdOx1 nCov-19: a catastrophic variant of vaccine-induced immune-mediated thrombotic thrombocytopenia: https:// ncbi.nlm.nih.gov/34341358/
- Cerebral venous sinus thrombosis associated with thrombocytopenia after COVID-19 vaccination: https://pubmed.ncbi.nlm.nih.gov/33845870/.
- central venous sinus thrombosis with subarachnoid hemorrhage after COVID-19 mRNA vaccination: are these reports merely coincidental: https:// ncbi.nlm.nih.gov/34478433/
- Cerebral venous sinus thrombosis negative for anti-PF4 antibody without thrombocytopenia after immunization with COVID-19 vaccine in a non-comorbid elderly Indian male treated with conventionalheparin-warfarin-based anticoagulation: https://pubmed.ncbi.nlm.nih.gov/34186376/
- Cerebral venous sinus thrombosis 2 weeks after first dose of SARS-CoV-2 mRNA vaccine: https://pubmed.ncbi.nlm.nih.gov/34101024/
- Deep venous thrombosis (DVT) occurring shortly after second dose of SARS-CoV-2 mRNA vaccine: https://pubmed.ncbi.nlm.nih.gov/33687691/
- Vaccine-induced immune thrombotic thrombocytopenia causing a severe form of cerebral venous thrombosis with high mortality rate: a case series: https:// ncbi.nlm.nih.gov/34393988/.
- Procoagulant microparticles: a possible link between vaccine-induced immune thrombocytopenia (VITT) and cerebral sinus venous thrombosis: https:// ncbi.nlm.nih.gov/34129181/.
- Acute cerebral venous thrombosis and pulmonary artery embolism associated with the COVID-19 vaccine: https://pubmed.ncbi.nlm.nih.gov/34247246/.
- Cerebral venous thrombosis following COVID-19 vaccination: https://ncbi.nlm.nih.gov/34045111/.
- Adverse effects reported after COVID-19 vaccination in a tertiary care hospital, centered on cerebral venous sinus thrombosis (CVST): https:// ncbi.nlm.nih.gov/34092166/
- Cerebral venous thrombosis associated with COVID-19 vaccine in Germany: https://ncbi.nlm.nih.gov/34288044/.
- Cerebral venous sinus thrombosis after COVID-19 vaccination : Neurological and radiological management: https://pubmed.ncbi.nlm.nih.gov/34327553/.
- Cerebral venous thrombosis and thrombocytopenia after COVID-19 vaccination: https://pubmed.ncbi.nlm.nih.gov/33878469/.
- Cerebral venous sinus thrombosis and thrombocytopenia after COVID-19 vaccination: report of two cases in the United Kingdom: https:// ncbi.nlm.nih.gov/33857630/.
- Cerebral venous thrombosis induced by SARS-CoV-2 vaccine: https://ncbi.nlm.nih.gov/34090750/.
- Cerebral venous sinus thrombosis associated with vaccine-induced thrombotic thrombocytopenia: https://pubmed.ncbi.nlm.nih.gov/34333995/
- Cerebral venous thrombosis after the BNT162b2 mRNA SARS-CoV-2 vaccine: https://pubmed.ncbi.nlm.nih.gov/34111775/.
- Cerebral venous thrombosis after COVID-19 vaccination: https://ncbi.nlm.nih.gov/34045111/
- Lethal cerebral venous sinus thrombosis after COVID-19 vaccination: https://ncbi.nlm.nih.gov/33983464/
- Cerebral venous sinus thrombosis in the U.S. population, After SARS-CoV-2 vaccination with adenovirusand after COVID-19: https://pubmed.ncbi.nlm.nih.gov/ 34116145/
- Cerebral venous thrombosis after COVID-19 vaccination: is the risk of thrombosis increased by intravascular administration of the vaccine: https:// ncbi.nlm.nih.gov/34286453/.
- Central venous sinus thrombosis with subarachnoid hemorrhage after COVID-19 mRNA vaccination: are these reports merely coincidental: https:// ncbi.nlm.nih.gov/34478433/
- Cerebral venous sinus thrombosis after ChAdOx1 nCov-19 vaccination with a misleading first brain MRI: https://pubmed.ncbi.nlm.nih.gov/34244448/
- Early results of bivalirudin treatment for thrombotic thrombocytopenia and cerebral venous sinus thrombosis after vaccination with Ad26.COV2.S: https:// ncbi.nlm.nih.gov/34226070/
- Cerebral venous sinus thrombosis associated with post-vaccination thrombocytopenia by COVID-19:https://pubmed.ncbi.nlm.nih.gov/33845870/.
- Cerebral venous sinus thrombosis 2 weeks after the first dose of SARS-CoV-2 mRNA vaccine: https://pubmed.ncbi.nlm.nih.gov/34101024/.
- Adverse effects reported after COVID-19 vaccination in a tertiary care hospital, focus on cerebral venous sinus thrombosis (CVST): https://pubmed.ncbi.nlm.nih.gov/ 34092166/
- Cerebral venous sinus thrombosis following vaccination against SARS-CoV-2: an analysis of cases reported to the European Medicines Agency: https:// ncbi.nlm.nih.gov/34293217/
- A rare case of a middle-age Asian male with cerebral venous thrombosis after COVID-19 AstraZenecavaccination: https://pubmed.ncbi.nlm.nih.gov/34274191/
- Massive cerebral venous thrombosis and venous basin infarction as late complications of COVID-19: a case report: https://pubmed.ncbi.nlm.nih.gov/ 34373991/
- Massive cerebral venous thrombosis due to vaccine-induced immune thrombotic thrombocytopenia: https://pubmed.ncbi.nlm.nih.gov/34261296/
- Cerebral venous thrombosis developing after COVID-19: VITT, VATT, TTS and more: https://pubmed.ncbi.nlm.nih.gov/34695859/
- Cerebral venous thrombosis and myeloproliferative neoplasms: a three-center study of 74 consecutive cases: https://pubmed.ncbi.nlm.nih.gov/34453762/.
- Vaccine-induced cerebral venous thrombosis and thrombocytopenia. Oxford- AstraZeneca COVID-19: amissed opportunity for rapid return on experience: https://sciencedirect.com/science/article/pii/S235255682100093X
- Case report: Take a second look: Cerebral venous thrombosis related to Covid-19 vaccination and thrombotic thrombocytopenia syndrome: https:// ncbi.nlm.nih.gov/34880826/
- ANCA-associated vasculitis after Pfizer-BioNTech COVID-19 vaccine: https://sciencedirect.com/science/article/pii/S0272638621007423
- propylthiouracil-induced neutrophil anti-cytoplasmic antibody-associated vasculitis after COVID-19 vaccination: https://pubmed.ncbi.nlm.nih.gov/34451967/
- IgA vasculitis in adult patient after vaccination with ChadOx1 nCoV-19: https://ncbi.nlm.nih.gov/34509658/
- A case of leukocytoclastic vasculitis after vaccination with a SARS-CoV2 vaccine: case report: https://pubmed.ncbi.nlm.nih.gov/34196469/
- A case of ANCA-associated vasculitis after AZD1222 (Oxford-AstraZeneca) SARS- CoV-2 vaccination:victim or causality?: https://pubmed.ncbi.nlm.nih.gov/34416184/.
- A case of ANCA-associated vasculitis after AZD1222 (Oxford-AstraZeneca) SARS- CoV-2 vaccination:victim or causality?: https://pubmed.ncbi.nlm.nih.gov/34416184/
- Leukocytoclastic vasculitis as a cutaneous manifestation of ChAdOx1 corona virus vaccine nCoV-19 (recombinant): https://pubmed.ncbi.nlm.nih.gov/34546608/
- Induction of cutaneous leukocytoclastic vasculitis after ChAdOx1 nCoV-19 vaccine: https://pubmed.ncbi.nlm.nih.gov/34853744/.
- Recurrent ANCA-associated vasculitis after Oxford AstraZeneca ChAdOx1-S COVID-19 vaccination: a case series of two patients: https:// ncbi.nlm.nih.gov/34755433/
- Cutaneous lymphocytic vasculitis after administration of the second dose of AZD1222 (Oxford-AstraZeneca) Severe acute respiratory syndrome Coronavirus 2 vaccine: chance or causality: https://pubmed.ncbi.nlm.nih.gov/34726187/.
- Case report: anti-neutrophil cytoplasmic antibody-associated vasculitis with acute renal failure and pulmonary hemorrhage can occur after COVID-19 vaccination: https://pubmed.ncbi.nlm.nih.gov/34859017/
- Intracerebral hemorrhage due to vasculitis following COVID-19 vaccination: case report: https://pubmed.ncbi.nlm.nih.gov/34783899/
- COVID-19 vaccine-induced urticarial vasculitis: https://pubmed.ncbi.nlm.nih.gov/ 34369046/.
- ANCA-associated vasculitis after Pfizer-BioNTech COVID-19 vaccine: https://ncbi.nlm.nih.gov/34280507/.
- New-onset leukocytoclastic vasculitis after COVID-19 vaccine: https://ncbi.nlm.nih.gov/34241833/
- Cutaneous small vessel vasculitis after COVID-19 vaccine: https://ncbi.nlm.nih.gov/34529877/.
- Outbreak of leukocytoclastic vasculitis after COVID-19 vaccine: https://ncbi.nlm.nih.gov/33928638/
- Leukocytoclastic vasculitis after exposure to COVID-19 vaccine: https://ncbi.nlm.nih.gov/34836739/
- Vasculitis and bursitis in [ 18 F] FDG-PET/CT after COVID-19 mRNA vaccine: post hoc ergo propter hoc?; https://pubmed.ncbi.nlm.nih.gov/34495381/.
- Cutaneous lymphocytic vasculitis after administration of COVID-19 mRNA vaccine:https://pubmed.ncbi.nlm.nih.gov/34327795
- Cutaneous leukocytoclastic vasculitis induced by Sinovac COVID-19 vaccine: https://ncbi.nlm.nih.gov/34660867/.
- Reactivation of IgA vasculitis after vaccination with COVID-19: https://ncbi.nlm.nih.gov/34848431/
- Varicella-zoster virus-related small-vessel vasculitis after Pfizer-BioNTech COVID-19 vaccination: https://pubmed.ncbi.nlm.nih.gov/34310759/.
- Imaging in vascular medicine: leukocytoclastic vasculitis after COVID-19 vaccine booster: https://pubmed.ncbi.nlm.nih.gov/34720009/
- Cutaneous vasculitis following COVID-19 vaccination: https://ncbi.nlm.nih.gov/34611627/.
- Possible case of COVID-19 mRNA vaccine-induced small-vessel vasculitis: https://ncbi.nlm.nih.gov/34705320/.
- IgA vasculitis following COVID-19 vaccination in an adult: https://ncbi.nlm.nih.gov/34779011/
- Propylthiouracil-induced anti-neutrophil cytoplasmic antibody-associated vasculitis following vaccination with COVID-19: https://pubmed.ncbi.nlm.nih.gov/34451967/
- Coronavirus disease vaccine 2019 (COVID-19) in systemic lupus erythematosus and neutrophil anti-cytoplasmic antibody-associated vasculitis: https:// ncbi.nlm.nih.gov/33928459/
- Reactivation of IgA vasculitis after COVID-19 vaccination: https://ncbi.nlm.nih.gov/34250509/
- First description of immune complex vasculitis after COVID-19 vaccination with BNT162b2: case report: https://pubmed.ncbi.nlm.nih.gov/34530771/.
- Nephrotic syndrome and vasculitis after SARS-CoV-2 vaccine: true association or circumstantial: https://pubmed.ncbi.nlm.nih.gov/34245294/.
- Occurrence of de novo cutaneous vasculitis after vaccination against coronavirus disease (COVID-19): https://pubmed.ncbi.nlm.nih.gov/34599716/.
- Asymmetric cutaneous vasculitis after COVID-19 vaccination with unusual preponderance ofeosinophils: https://pubmed.ncbi.nlm.nih.gov/34115904/.
- Granulomatous vasculitis after AstraZeneca anti-SARS-CoV-2 vaccine: https://ncbi.nlm.nih.gov/34237323/.
- A case of generalized Sweet’s syndrome with vasculitis triggered by recent vaccination with COVID-19: https://pubmed.ncbi.nlm.nih.gov/34849386/
- Small-vessel vasculitis following Oxford-AstraZeneca vaccination against SARS- CoV-2: https://pubmed.ncbi.nlm.nih.gov/34310763/
- Cutaneous vasculitis after severe acute respiratory syndrome coronavirus 2 vaccine: https://pubmed.ncbi.nlm.nih.gov/34557622/.
- Leukocytoclastic vasculitis after coronavirus disease vaccination 2019: https://ncbi.nlm.nih.gov/34713472/803
- Outbreaks of mixed cryoglobulinemia vasculitis after vaccination against SARS- CoV-2: https://pubmed.ncbi.nlm.nih.gov/34819272/
- Cutaneous small-vessel vasculitis after vaccination with a single dose of Janssen Ad26.COV2.S: https://pubmed.ncbi.nlm.nih.gov/34337124/
- Case of immunoglobulin A vasculitis after vaccination against coronavirus disease 2019: https://pubmed.ncbi.nlm.nih.gov/34535924/
- Relapse of microscopic polyangiitis after COVID-19 vaccination: case report: https://ncbi.nlm.nih.gov/34251683/.
- GM1 ganglioside antibody and COVID-19-related Guillain Barre syndrome: case report, systemic review, and implications for vaccine development: https://sciencedirect.com/science/article/pii/S2666354621000065
- Guillain-Barré syndrome after AstraZeneca COVID-19 vaccination: causal or casual association:https://sciencedirect.com/science/article/pii/S0303846721004169
- Sensory Guillain-Barré syndrome after ChAdOx1 nCov-19 vaccine: report of two cases and review of theliterature: https://sciencedirect.com/science/article/pii/ S0165572821002186
- Guillain-Barré syndrome after the first dose of SARS-CoV-2 vaccine: a temporary occurrence, not a causalassociation: https://sciencedirect.com/science/article/ pii/S2214250921000998.
- Guillain-Barré syndrome presenting as facial diplegia after vaccination with COVID-19: a casereport: https://sciencedirect.com/science/article/pii/ S0736467921006442
- Guillain-Barré syndrome after the first injection of ChAdOx1 nCoV-19 vaccine: first report: https://sciencedirect.com/science/article/pii/S0035378721005853.
- SARS-CoV-2 vaccines are not safe for those with Guillain-Barre syndrome following vaccination: https://sciencedirect.com/science/article/pii/S2049080121005343
- Guillian Barré syndrome after vaccination with mRNA-1273 against COVID-19: https://pubmed.ncbi.nlm.nih.gov/34477091/
- A novel case of bifacial diplegia variant of Guillain-Barré syndrome after vaccination with Janssen COVID-19: https://pubmed.ncbi.nlm.nih.gov/34449715/
- Sensory Guillain-Barré syndrome following ChAdOx1 nCov-19 vaccine: report of two cases and review of the literature: https://pubmed.ncbi.nlm.nih.gov/34416410/.
- Facial diplegia: a rare and atypical variant of Guillain-Barré syndrome and the Ad26.COV2.S vaccine: https://pubmed.ncbi.nlm.nih.gov/34447646/
- Guillain-Barré syndrome after ChAdOx1 nCoV-19 COVID-19 vaccination: a case series: https://pubmed.ncbi.nlm.nih.gov/34548920/
- AstraZeneca COVID-19 vaccine and Guillain-Barré syndrome in Tasmania: a causal link: https://pubmed.ncbi.nlm.nih.gov/34560365/
- COVID-19, Guillain-Barré and vaccineA dangerous mix: https://ncbi.nlm.nih.gov/34108736/.
- Guillain-Barré syndrome after the first dose of Pfizer-BioNTech COVID-19 vaccine: case report and review of reported cases: https://pubmed.ncbi.nlm.nih.gov/ 34796417/.
- Guillain-Barre syndrome after BNT162b2 COVID-19 vaccine: https://springer.com/article/10.1007%2Fs10072-021-05523-5.
- COVID-19 adenovirus vaccines and Guillain-Barré syndrome with facial palsy: https://onlinelibrary.wiley.com/doi/10.1002/ana.26258.
- Association of receipt association of Ad26.COV2.S COVID-19 vaccine with presumed Guillain-Barresyndrome, February-July 2021: https://jamanetwork.com/ journals/jama/fullarticle/2785009
- A case of Guillain-Barré syndrome after Pfizer COVID-19 vaccine: https://ncbi.nlm.nih.gov/34567447/
- Guillain-Barré syndrome associated with COVID-19 vaccination: https://ncbi.nlm.nih.gov/34648420/.
- Rate of recurrent Guillain-Barré syndrome after COVID-19 BNT162b2 mRNA vaccine: https://jamanetwork.com/journals/jamaneurology/fullarticle/2783708
- Guillain-Barre syndrome after COVID-19 vaccination in an adolescent: https://pedneur.com/article/S0887-8994(21)00221-6/fulltext.
- Guillain-Barre syndrome after ChAdOx1-S / nCoV-19 vaccination: https://ncbi.nlm.nih.gov/34114256/.
- Guillain-Barre syndrome after COVID-19 mRNA-1273 vaccine: case report: https://ncbi.nlm.nih.gov/34767184/.
- Guillain-Barre syndrome following SARS-CoV-2 vaccination in 19 patients: https://ncbi.nlm.nih.gov/34644738/.
- Guillain-Barre syndrome presenting with facial diplegia following vaccination with COVID-19 in two patients: https://pubmed.ncbi.nlm.nih.gov/34649856/
- A rare case of Guillain-Barré syndrome after COVID-19 vaccination: https://ncbi.nlm.nih.gov/34671572/
- Neurological complications of COVID-19: Guillain-Barre syndrome after Pfizer COVID-19 vaccine: https://pubmed.ncbi.nlm.nih.gov/33758714/
- COVID-19 vaccine causing Guillain-Barre syndrome, an uncommon potential side effect: https://pubmed.ncbi.nlm.nih.gov/34484780/
- Guillain-Barre syndrome after the first dose of COVID-19 vaccination: case report; https://pubmed.ncbi.nlm.nih.gov/34779385/.
- Guillain-Barre syndrome after the first injection of ChAdOx1 nCoV-19 vaccine: first report: https://pubmed.ncbi.nlm.nih.gov/34217513/.
- A case of sensory ataxic Guillain-Barre syndrome with immunoglobulin G anti-GM1 antibodies after first dose of COVID-19 BNT162b2 mRNA vaccine (Pfizer): https:// ncbi.nlm.nih.gov/34871447/
- A variant of Guillain-Barré syndrome after SARS-CoV-2 vaccination: AMSAN: https://ncbi.nlm.nih.gov/34370408/.
- A rare variant of Guillain-Barré syndrome after vaccination with COV2.S:https://pubmed.ncbi.nlm.nih.gov/34703690/.
- Guillain-Barré syndrome after SARS-CoV-2 vaccination in a patient with previous vaccine-associated Guillain-Barré syndrome: https://pubmed.ncbi.nlm.nih.gov/ 34810163/
- Guillain-Barré syndrome in an Australian state using mRNA and adenovirus-vector SARS-CoV-2 vaccines: https://onlinelibrary.wiley.com/doi/10.1002/ana.26218
- Variant Guillain-Barré syndrome occurring after SARS-CoV-2 vaccination: https://ncbi.nlm.nih.gov/34114269/
- Guillian-Barre syndrome with axonal variant temporally associated with Modern SARS-CoV-2 mRNA-based vaccine: https://pubmed.ncbi.nlm.nih.gov/34722067/
- Guillain-Barre syndrome after the first dose of SARS-CoV-2 vaccine: a temporary occurrence, not acausal association: https://pubmed.ncbi.nlm.nih.gov/33968610/
- SARS-CoV-2 vaccines can be complicated not only by Guillain-Barré syndrome but also by distal small fiber neuropathy: https://pubmed.ncbi.nlm.nih.gov/34525410/
- Clinical variant of Guillain-Barré syndrome with prominent facial diplegia after AstraZeneca 2019coronavirus disease vaccine: https://pubmed.ncbi.nlm.nih.gov/ 34808658/
- Miller-Fisher syndrome and Guillain-Barré syndrome overlap syndrome in a patient after Oxford-AstraZeneca SARS-CoV-2 vaccination: https://pubmed.ncbi.nlm.nih.gov/ 34848426/
- Bilateral facial weakness with a variant of paresthesia of Guillain-Barre syndrome after Vaxzevria COVID-19 vaccine: https://pubmed.ncbi.nlm.nih.gov/34261746/
- Rare case of contralateral supraclavicular lymphadenopathy after vaccination with COVID-19: computed tomography and ultrasound findings: https:// ncbi.nlm.nih.gov/34667486/
- COVID-19 mRNA vaccination-induced lymphadenopathy mimics lymphoma progression on FDG PET / CT: https://pubmed.ncbi.nlm.nih.gov/33591026/
- Lymphadenopathy in COVID-19 vaccine recipients: diagnostic dilemma in oncology patients: https://pubmed.ncbi.nlm.nih.gov/33625300/
- Hypermetabolic lymphadenopathy after administration of BNT162b2 mRNA vaccine Covid-19: incidence assessed by [ 18 F] FDG PET-CT and relevance for study interpretation: https://pubmed.ncbi.nlm.nih.gov/33774684/
- Lymphadenopathy after COVID-19 vaccination: review of imaging findings: https://ncbi.nlm.nih.gov/33985872/
- Lymphadenopathy associated with COVID-19 vaccination on FDG PET/CT: distinguishing features in adenovirus-vectored vaccine: https:// ncbi.nlm.nih.gov/34115709/.
- COVID-19 vaccination-induced lymphadenopathy in a specialized breast imaging clinic in Israel: analysisof 163 cases: https://pubmed.ncbi.nlm.nih.gov/34257025/.
- Coronavirus disease vaccine 2019 mimics lymph node metastases in patients undergoing skin cancer follow-up: a single-center study: https:// ncbi.nlm.nih.gov/34280870/
- COVID-19 post-vaccination lymphadenopathy: report of fine-needle aspiration biopsy cytologic findings: https://pubmed.ncbi.nlm.nih.gov/34432391/
- Regional lymphadenopathy after COVID-19 vaccination: review of the literature and considerations for patient management in breast cancer care: https:// ncbi.nlm.nih.gov/34731748/
- Adverse events of COVID injection that may occur in children.Acute-onset supraclavicularlymphadenopathy coincident with intramuscular mRNA vaccination against COVID-19 may be related to the injection technique of the vaccine, Spain, January and February 2021: https://pubmed.ncbi.nlm.nih.gov/33706861/
- Supraclavicular lymphadenopathy after COVID-19 vaccination in Korea: serial follow- up by ultrasonography: https://pubmed.ncbi.nlm.nih.gov/34116295/
- Oxford-AstraZeneca COVID-19 vaccination induced lymphadenopathy on [18F] choline PET / CT, not just an FDG finding: https://pubmed.ncbi.nlm.nih.gov/ 33661328/
- A case of cervical lymphadenopathy following COVID-19 vaccination: https://ncbi.nlm.nih.gov/34141500/
- Unilateral lymphadenopathy after COVID-19 vaccination: a practical management plan for radiologists of all specialties: https://pubmed.ncbi.nlm.nih.gov/33713605/
- Supraclavicular lymphadenopathy after COVID-19 vaccination: an increasing presentation in the two-weekwait neck lump clinic: https://pubmed.ncbi.nlm.nih.gov/ 33685772/
- COVID-19 vaccination and lower cervical lymphadenopathy in two-week neck lump clinic: a follow-up audit: https://pubmed.ncbi.nlm.nih.gov/33947605/.
- Cervical lymphadenopathy after coronavirus disease vaccination 2019: clinical features and implications for head and neck cancer services: https:// ncbi.nlm.nih.gov/34526175/
- Lymphadenopathy associated with the COVID-19 vaccine: https://ncbi.nlm.nih.gov/33786231/
- Evolution of lymphadenopathy on PET/MRI
- Massive cervical lymphadenopathy following vaccination with COVID-19: https://ncbi.nlm.nih.gov/34601889/
- Acute-onset supraclavicular lymphadenopathy coincident with intramuscular mRNA vaccination against COVID-19 may be related to the injection technique of the vaccine, Spain, January and February 2021: https://pubmed.ncbi.nlm.nih.gov/ 33706861/
- Supraclavicular lymphadenopathy after COVID-19 vaccination in Korea: serial follow- up by ultrasonography: https://pubmed.ncbi.nlm.nih.gov/34116295/
- Oxford-AstraZeneca COVID-19 vaccination induced lymphadenopathy on [18F] choline PET / CT, not just an FDG finding: https://pubmed.ncbi.nlm.nih.gov/ 33661328/
- A case of cervical lymphadenopathy following COVID-19 vaccination: https://ncbi.nlm.nih.gov/34141500/
- Unilateral lymphadenopathy after COVID-19 vaccination: a practical management plan for radiologists of all specialties: https://pubmed.ncbi.nlm.nih.gov/33713605/
- Supraclavicular lymphadenopathy after COVID-19 vaccination: an increasing presentation in the two-weekwait neck lump clinic: https://pubmed.ncbi.nlm.nih.gov/ 33685772/
- COVID-19 vaccination and lower cervical lymphadenopathy in two-week neck lump clinic: a follow-up audit: https://pubmed.ncbi.nlm.nih.gov/33947605/.
- Cervical lymphadenopathy after coronavirus disease vaccination 2019: clinical features and implications for head and neck cancer services: https:// ncbi.nlm.nih.gov/34526175/
- Lymphadenopathy associated with the COVID-19 vaccine: https://ncbi.nlm.nih.gov/33786231/
- Evolution of lymphadenopathy on PET/MRI after COVID-19 vaccination: https://ncbi.nlm.nih.gov/33625301/.
- Massive cervical lymphadenopathy following vaccination with COVID-19: https://ncbi.nlm.nih.gov/34601889/
- COVID-19 vaccine-related axillary and cervical lymphadenopathy in patients with current or previousbreast cancer and other malignancies: cross-sectional imaging findings on MRI, CT and PET-CT: https://pubmed.ncbi.nlm.nih.gov/34719892/
- Supraclavicular lymphadenopathy after COVID-19 vaccination in Korea: serial follow- up by ultrasonography: https://pubmed.ncbi.nlm.nih.gov/34116295/.
- Evolution of Lymphadenopathy at Pet/MRI after COVID-19 vaccination: https://ncbi.nlm.nih.gov/33625301/
- COVID-19 vaccine-associated anaphylaxis: a statement from the Anaphylaxis Committee of the WorldAllergy Organization:. https://sciencedirect.com/ science/article/pii/S1939455121000119.
- Allergic reactions, including anaphylaxis, after receiving the first dose of the Pfizer- BioNTech COVID-19 vaccine: https://pubmed.ncbi.nlm.nih.gov/33475702/
- Allergic reactions, including anaphylaxis, after receiving the first dose of Pfizer- BioNTech COVID-19 vaccine – United States, December 14-23, 2020: https:// ncbi.nlm.nih.gov/33444297/
- Allergic reactions, including anaphylaxis, after receiving first dose of Modern COVID-19 vaccine – UnitedStates, December 21, 2020-January 10, 2021: https:// ncbi.nlm.nih.gov/33507892/
- Reports of anaphylaxis after coronavirus disease vaccination 2019, South Korea, February 26-April 30, 2021: https://pubmed.ncbi.nlm.nih.gov/34414880/
- Reports of anaphylaxis after receiving COVID-19 mRNA vaccines in the S.-Dec 14, 2020-Jan 18, 2021: https://pubmed.ncbi.nlm.nih.gov/33576785/
- Immunization practices and risk of anaphylaxis: a current, comprehensive update of COVID-19 vaccination data: https://pubmed.ncbi.nlm.nih.gov/34269740/
- Relationship between pre-existing allergies and anaphylactic reactions following administration of COVID-19 mRNA vaccine: https://pubmed.ncbi.nlm.nih.gov/ 34215453/
- Anaphylaxis Associated with COVID-19 mRNA Vaccines: Approach to Allergy Research: https://pubmed.ncbi.nlm.nih.gov/33932618/
- Allergic reactions and anaphylaxis to LNP-based COVID-19 vaccines: https://ncbi.nlm.nih.gov/33571463/
- Cumulative adverse event report of anaphylaxis following injections of COVID-19 mRNA vaccine (Pfizer-BioNTech) in Japan: the first month report: https:// ncbi.nlm.nih.gov/34347278/
- COVID-19 vaccines increase the risk of anaphylaxis: https://ncbi.nlm.nih.gov/33685103/
- Biphasic anaphylaxis after exposure to the first dose of the Pfizer-BioNTech COVID-19 mRNA vaccineCOVID-19: https://pubmed.ncbi.nlm.nih.gov/34050949/
- Polyethylene glycol (PEG) is a cause of anaphylaxis to Pfizer / BioNTech mRNA COVID-19 vaccine: https://pubmed.ncbi.nlm.nih.gov/33825239/
- Elevated rates of anaphylaxis after vaccination with Pfizer BNT162b2 mRNA vaccine against COVID-19 in Japanese healthcare workers; a secondary analysis of initial post-approval safety data: https://pubmed.ncbi.nlm.nih.gov/34128049/
- .IgE-mediated allergy to polyethylene glycol (PEG) as a cause of anaphylaxis to COVID-19 mRNA vaccines: https://pubmed.ncbi.nlm.nih.gov/34318537/
- Anaphylactic reactions to COVID-19 mRNA vaccines: a call for further studies: https://pubmed.ncbi.nlm.nih.gov/33846043/ 188.
- Anaphylaxis following Covid-19 vaccine in a patient with cholinergic urticaria: https://ncbi.nlm.nih.gov/33851711/
- Anaphylaxis induced by CoronaVac COVID-19 vaccine: clinical features and results of revaccination: https://pubmed.ncbi.nlm.nih.gov/34675550/.
- Anaphylaxis after Modern COVID-19 vaccine: https://pubmed.ncbi.nlm.nih.gov/ 34734159/
- Sex differences in the incidence of anaphylaxis to LNP-mRNA vaccines COVID-19: https://pubmed.ncbi.nlm.nih.gov/34020815/
- Allergic reactions, including anaphylaxis, after receiving the first dose of Pfizer- BioNTech COVID-19vaccine – United States, December 14 to 23, 2020: https:// ncbi.nlm.nih.gov/33641264/
- Allergic reactions, including anaphylaxis, after receiving the first dose of Modern COVID-19 vaccine –United States, December 21, 2020 to January 10, 2021: https:// ncbi.nlm.nih.gov/33641268/
- Prolonged anaphylaxis to Pfizer 2019 coronavirus disease vaccine: a case report and mechanism of action: https://pubmed.ncbi.nlm.nih.gov/33834172/
- Anaphylaxis reactions to Pfizer BNT162b2 vaccine: report of 3 cases of anaphylaxis following vaccination with Pfizer BNT162b2: https://pubmed.ncbi.nlm.nih.gov/ 34579211/
- Biphasic anaphylaxis after first dose of 2019 messenger RNA coronavirus disease vaccine with positivepolysorbate 80 skin test result: https://pubmed.ncbi.nlm.nih.gov/ 34343674/
- Biphasic anaphylaxis after exposure to the first dose of Pfizer-BioNTech COVID-19 mRNA vaccine COVID-19: https://pubmed.ncbi.nlm.nih.gov/34050949/
- Biphasic anaphylaxis after exposure to the first dose of Pfizer-BioNTech COVID-19 mRNA vaccine COVID-19: https://pubmed.ncbi.nlm.nih.gov/34050949/
- Iguchi, , Umeda, H., Kojima, M., Kanno, Y., Tanaka, Y., Kinoshita, N., & Sato, D. (2021). CumulativeAdverse Event Reporting of Anaphylaxis After mRNA COVID-19 Vaccine (Pfizer-BioNTech) Injections in Japan: The First-Month Report. Drug Saf, 44(11), 1209-1214. doi:10.1007/s40264-021-01104-9.https://www.ncbi.nlm.nih.gov/ pubmed/34347278
- Team, C.-R., Food, & Drug, A. (2021). Allergic Reactions Including Anaphylaxis After Receipt of the First Dose of Pfizer-BioNTech COVID-19 Vaccine – United States, December 14-23, 2020. MMWR Morb Mortal Wkly Rep, 70(2), 46-51. doi: 10.15585/mmwr.mm7002e1. https://www.ncbi.nlm.nih.gov/pubmed/33444297
- Myopericarditis after Pfizer mRNA COVID-19 vaccination in adolescents: https://sciencedirect.com/science/article/pii/S002234762100665X
- Myopericarditis after vaccination with COVID-19 mRNA in adolescents 12 to 18 years of age: https://sciencedirect.com/science/article/pii/S0022347621007368
- Important information on myopericarditis after vaccination with Pfizer COVID-19 mRNA in adolescents: https://sciencedirect.com/science/article/pii/ S0022347621007496
- Insights from a murine model of COVID-19 mRNA vaccine-induced myopericarditis: could accidental intravenous injection of a vaccine induce myopericarditis https:// oup.com/cid/advance-article/doi/10.1093/cid/ciab741/6359059
- Acute myocarditis after administration of BNT162b2 vaccine against COVID-19:. https://sciencedirect.com/science/article/abs/pii/S188558572100133X
- Insights from a murine model of myopericarditis induced by COVID-19 mRNA vaccine: could accidentalintravenous injection of a vaccine induce myopericarditis: https://pubmed.ncbi.nlm.nih.gov/34453510/
- COVID-19 mRNA vaccination and development of CMR-confirmed myopericarditis: https://medrxiv.org/content/10.1101/2021.09.13.21262182v1.full?s=09.
- Intravenous injection of coronavirus disease 2019 (COVID-19) mRNA vaccine can induce acute myopericarditis in a mouse model: https://t.co/j0IEM8cMXI
- Myopericarditis in a previously healthy adolescent male after COVID-19 vaccination: Case report: https://pubmed.ncbi.nlm.nih.gov/34133825/
- Report of a case of myopericarditis after vaccination with BNT162b2 COVID-19 mRNA in a young Korean male: https://pubmed.ncbi.nlm.nih.gov/34636504/
- Myopericarditis after Pfizer messenger ribonucleic acid coronavirus coronavirus disease vaccine in adolescents: https://pubmed.ncbi.nlm.nih.gov/34228985/
- Acute myopericarditis after COVID-19 vaccine in adolescents: https://ncbi.nlm.nih.gov/34589238/
- Nygaard, U., Holm, M., Bohnstedt, C., Chai, Q., Schmidt, L. S., Hartling, U. B., . . . Stensballe, L. G. (2022). Population-based Incidence of Myopericarditis After COVID-19 Vaccination in Danish Pediatr Infect Dis J, 41(1), e25-e28. doi:10.1097/INF.0000000000003389. https://www.ncbi.nlm.nih.gov/pubmed/ 34889875
- Schauer, J., Buddhe, S., Colyer, J., Sagiv, E., Law, , Mallenahalli Chikkabyrappa, S., & Portman, M. A.(2021). Myopericarditis After the Pfizer Messenger Ribonucleic Acid Coronavirus Disease Vaccine inAdolescents. J Pediatr, 238, 317-320. doi: 10.1016/j.jpeds.2021.06.083. https://www.ncbi.nlm.nih.gov/pubmed/34228985
- Kohli, , Desai, L., Chowdhury, D., Harahsheh, A. S., Yonts, A. B., Ansong, A., . . . Ang, J. Y. (2021). mRNA Coronavirus-19 Vaccine-Associated Myopericarditis in Adolescents: A Survey Study. J Pediatr. doi:10.1016/j.jpeds.2021.12.025. https:// www.ncbi.nlm.nih.gov/pubmed/34952008
- Long, S. (2021). Important Insights into Myopericarditis after the Pfizer mRNA COVID-19 Vaccination in Adolescents. J Pediatr, 238, 5. doi:10.1016/j.jpeds. 2021.07.057. https://www.ncbi.nlm.nih.gov/pubmed/34332972
- McLean, K., & Johnson, T. J. (2021). Myopericarditis in a previously healthy adolescent male followingCOVID-19 vaccination: A case Acad Emerg Med, 28(8), 918-921. doi:10.1111/acem.14322. https://www.ncbi.nlm.nih.gov/pubmed/
34133825
- Umei, C., Kishino, Y., Shiraishi, Y., Inohara, T., Yuasa, S., & Fukuda, K. (2021). Recurrence of myopericarditis following mRNA COVID-19 vaccination in a male adolescent. CJC Open.doi:10.1016/j.cjco.2021.12.002. https://www.ncbi.nlm.nih.gov/ pubmed/34904134
- Das, B., Kohli, U., Ramachandran, P., Nguyen, H. H., Greil, G., Hussain, T., . . . Khan, D. (2021).Myopericarditis after messenger RNA Coronavirus Disease 2019 Vaccination in Adolescents 12 to 18 Years of Age. J Pediatr, 238, 26-32 e21. doi: 10.1016/j.jpeds.2021.07.044. https://www.ncbi.nlm.nih.gov/pubmed/34339728
- Gatti, M., Raschi, E., Moretti, U., Ardizzoni, , Poluzzi, E., & Diemberger, I. (2021). Influenza Vaccinationand Myo-Pericarditis in Patients Receiving Immune Checkpoint
Inhibitors: Investigating the Likelihood of Interaction through the Vaccine Adverse Event Reporting System and VigiBase. Vaccines (Basel), 9(1). doi:10.3390/ vaccines9010019. https://www.ncbi.nlm.nih.gov/pubmed/33406694
- Myopericarditis in a previously healthy adolescent male after COVID-19 vaccination: Case report: https://pubmed.ncbi.nlm.nih.gov/34133825/
- An academic hospital experience assessing the risk of COVID-19 mRNA vaccine using patient’s allergy history: https://sciencedirect.com/science/article/pii/ S2213219821007972
- Allergic reactions, including anaphylaxis, after receiving the first dose of the Pfizer- BioNTech COVID-19 vaccine: https://pubmed.ncbi.nlm.nih.gov/33475702/
- Allergic reactions to the first COVID-19 vaccine: a potential role of polyethylene glycol: https://pubmed.ncbi.nlm.nih.gov/33320974/
- Pfizer Vaccine Raises Allergy Concerns: https://pubmed.ncbi.nlm.nih.gov/33384356/
- Allergic reactions, including anaphylaxis, after receiving the first dose of Pfizer- BioNTech COVID-19 vaccine – United States, December 14-23, 2020: https:// ncbi.nlm.nih.gov/33444297/
- Allergic reactions, including anaphylaxis, after receiving first dose of Modern COVID-19 vaccine – UnitedStates, December 21, 2020-January 10, 2021: https:// ncbi.nlm.nih.gov/33507892/
- Severe Allergic Reactions after COVID-19 Vaccination with the Pfizer / BioNTech Vaccine in Great Britain and the USA: Position Statement of the German Allergy Societies: German Medical Association ofAllergologists (AeDA), German Society for Allergology and Clinical Immunology (DGAKI) and Society for Pediatric Allergology and Environmental Medicine (GPA): https://pubmed.ncbi.nlm.nih.gov/33643776/
- Allergic reactions and anaphylaxis to LNP-based COVID-19 vaccines: https://ncbi.nlm.nih.gov/33571463/
- Allergenic components of the mRNA-1273 vaccine for COVID-19: possible involvement of polyethyleneglycol and IgG-mediated complement activation: https:// ncbi.nlm.nih.gov/33657648/
- Acute allergic reactions to COVID-19 mRNA vaccines: https://ncbi.nlm.nih.gov/33683290/
- Polyethylene glycole allergy of the SARS CoV2 vaccine recipient: case report of a young adult recipientand management of future exposure to SARS-CoV2: https:// ncbi.nlm.nih.gov/33919151/
- Allergic reactions and adverse events associated with administration of mRNA-based vaccines. A health system experience: https://pubmed.ncbi.nlm.nih.gov/34474708/
- Allergic reactions to COVID-19 vaccines: statement of the Belgian Society of Allergy and Clinical Immunology (BelSACI): https://tandfonline.com/doi/abs/ 10.1080/17843286.2021.1909447
- Allergic reactions after COVID-19 vaccination: putting the risk in perspective: https://ncbi.nlm.nih.gov/34463751/
- Risk of severe allergic reactions to COVID-19 vaccines among patients with allergic skin disease: practical An ETFAD position statement with external experts: https://pubmed.ncbi.nlm.nih.gov/33752263/
- Association of self-reported history of high-risk allergy with allergy symptoms after COVID-19 vaccination: https://pubmed.ncbi.nlm.nih.gov/34698847/
- Greenhawt, M., Abrams, E. M., Shaker, M., Chu, D. K., Khan, D., Akin, C., . . . Golden, B. K. (2021). The Risk of Allergic Reaction to SARS-CoV-2 Vaccines and Recommended Evaluation and Management: A Systematic Review, Meta-Analysis, GRADE Assessment, and International Consensus Approach. J AllergyClin Immunol Pract, 9(10), 3546-3567. doi:10.1016/j.jaip.2021.06.006. https://www.ncbi.nlm.nih.gov/pubmed/34153517
- Klimek, , Bergmann, K. C., Brehler, R., Pfutzner, W., Zuberbier, T., Hartmann, K., . .
. Worm, M. (2021). Practical handling of allergic reactions to COVID-19 vaccines: A position paper fromGerman and Austrian Allergy Societies AeDA, DGAKI, GPA and OGAI. Allergo J Int, 1-17. doi:10.1007/s40629-021-00165-7. https:// www.ncbi.nlm.nih.gov/pubmed/33898162
- Klimek, L., Novak, N., Hamelmann, E., Werfel, T., Wagenmann, M., Taube, C., . . . Worm, M. (2021). Severe allergic reactions after COVID-19 vaccination with the Pfizer/BioNTech vaccine in Great Britain and USA:Position statement of the German Allergy Societies: Medical Association of German Allergologists (AeDA), German Society for Allergology and Clinical Immunology (DGAKI) and Society for Pediatric Allergology and Environmental Medicine (GPA). Allergo J Int, 30(2), 51-55. doi: 10.1007/s40629-020-00160-4. https://ncbi.nlm.nih.gov/pubmed/33643776
- Warren, C. M., Snow, T., Lee, A. S., Shah, M. M., Heider, A., Blomkalns, A., . . . Nadeau, K. C. (2021).Assessment of Allergic and Anaphylactic Reactions to mRNA COVID-19 Vaccines With Confirmatory Testing in a US Regional Health System. JAMA Netw Open, 4(9), e2125524. doi:10.1001/jamanetworkopen.2021.25524. https://www.ncbi.nlm.nih.gov/pubmed/34533570
- Bell’s palsy and SARS-CoV-2 vaccines: an unfolding story: https://sciencedirect.com/science/article/pii/S1473309921002735
- Bell’s palsy after the second dose of the Pfizer COVID-19 vaccine in a patient with a history of recurrent Bell’s palsy: https://sciencedirect.com/science/article/pii/ S266635462100020X
- Bell’s palsy after COVID-19 vaccination: case report: https://sciencedirect.com/science/article/pii/S217358082100122X.
- The association between COVID-19 vaccination and Bell’s palsy: https://ncbi.nlm.nih.gov/34411533/
- Bell’s palsy after COVID-19 vaccination: https://pubmed.ncbi.nlm.nih.gov/33611630/
- Bell’s palsy after 24 hours of mRNA-1273 SARS-CoV-2 mRNA-1273 vaccine: https://ncbi.nlm.nih.gov/34336436/
- Bell’s palsy after COV2.S COVID-19 vaccination: https://pubmed.ncbi.nlm.nih.gov/34014316/
- Bell’s palsy after COVID-19 vaccination: case report: https://ncbi.nlm.nih.gov/34330676/
- Acute facial paralysis as a possible complication of SARS-CoV-2 vaccination: https://ncbi.nlm.nih.gov/33975372/.
- Bell’s palsy after COVID-19 vaccination with high antibody response in CSF: https://ncbi.nlm.nih.gov/34322761/.
- Bell’s palsy after a single dose of vaccine SARS-CoV-2: case report: https://pubmed.ncbi.nlm.nih.gov/34032902/.
- Adverse event reporting and risk of Bell’s palsy after COVID-19 vaccination: https://thelancet.com/journals/laninf/article/PIIS1473-3099(21)00646-0/fulltext.
- Bilateral facial nerve palsy and COVID-19 vaccination: causality or coincidence:https://pubmed.ncbi.nlm.nih.gov/34522557/
- Left Bell’s palsy after the first dose of mRNA-1273 SARS-CoV-2 vaccine: case report:https://pubmed.ncbi.nlm.nih.gov/34763263/.
- Bell’s palsy after inactivated vaccination with COVID-19 in a patient with a history of recurrent Bell’s palsy: case report: https://pubmed.ncbi.nlm.nih.gov/34621891/
- Bell’s palsy after vaccination with mRNA (BNT162b2) and inactivated (CoronaVac) SARS-CoV-2 vaccines: a case series and a nested case-control study: https:// ncbi.nlm.nih.gov/34411532/
- A case of acute demyelinating polyradiculoneuropathy with bilateral facial palsy after ChAdOx1 nCoV-19 vaccine:. https://pubmed.ncbi.nlm.nih.gov/34272622/
- Type I interferons as a potential mechanism linking COVID-19 mRNA vaccines with Bell’s palsy: https://pubmed.ncbi.nlm.nih.gov/33858693/
- COVID-19 vaccine-induced axillary and pectoral lymphadenopathy in PET: https://sciencedirect.com/science/article/pii/S1930043321002612
- Evolution of bilateral hypermetabolic axillary hypermetabolic lymphadenopathy on FDG PET/CT after 2-dose COVID-19 vaccination: https://pubmed.ncbi.nlm.nih.gov/ 34735411/
- COVID-19 vaccine-related axillary lymphadenopathy in breast cancer patients: case series with literature review: https://pubmed.ncbi.nlm.nih.gov/34836672/.
- Subclinical axillary lymphadenopathy associated with COVID-19 vaccination on screening mammography: https://pubmed.ncbi.nlm.nih.gov/34906409/
- Axillary adenopathy associated with COVID-19 vaccination: imaging findings and follow-up recommendations in 23 women: https://pubmed.ncbi.nlm.nih.gov/ 33624520/
- Unilateral axillary adenopathy in the setting of COVID-19 vaccination: follow-up: https://pubmed.ncbi.nlm.nih.gov/34298342/
- COVID-19 vaccine-related axillary and cervical lymphadenopathy in patients with current or previousbreast cancer and other malignancies: cross-sectional imaging findings on MRI, CT and PET-CT: https://pubmed.ncbi.nlm.nih.gov/34719892/
- Incidence of axillary adenopathy on breast imaging after vaccination with COVID-19:https://pubmed.ncbi.nlm.nih.gov/34292295/.
- Unilateral axillary lymphadenopathy related to COVID-19 vaccine: pattern on screening breast MRIallowing benign evaluation: https://pubmed.ncbi.nlm.nih.gov/ 34325221/
- Axillary lymphadenopathy in patients with recent Covid-19 vaccination: a new diagnostic dilemma: https://pubmed.ncbi.nlm.nih.gov/34825530/.
- COVID-19 vaccine-induced unilateral axillary adenopathy: follow-up evaluation in the USA: https://pubmed.ncbi.nlm.nih.gov/34655312/.
- Axillary adenopathy associated with COVID-19 vaccination: imaging findings and follow-up recommendations in 23 women: https://pubmed.ncbi.nlm.nih.gov/ 33624520/
- Unilateral axillary adenopathy in the setting of COVID-19 vaccination: follow-up: https://pubmed.ncbi.nlm.nih.gov/34298342/
- Incidence of axillary adenopathy on breast imaging after vaccination with COVID-19:https://pubmed.ncbi.nlm.nih.gov/34292295/.
- Unilateral axillary lymphadenopathy related to COVID-19 vaccine: pattern on screening breast MRIallowing benign evaluation: https://pubmed.ncbi.nlm.nih.gov/ 34325221/
- Axillary lymphadenopathy in patients with recent Covid-19 vaccination: a new diagnostic dilemma: https://pubmed.ncbi.nlm.nih.gov/34825530/.
- COVID-19 vaccine-induced unilateral axillary adenopathy: follow-up evaluation in the USA: https://pubmed.ncbi.nlm.nih.gov/34655312/.
- Adenopathy after COVID-19 vaccination: https://pubmed.ncbi.nlm.nih.gov/ 33625299/.
- Myocarditis and pericarditis after vaccination with COVID-19 mRNA: practical considerations for careproviders: https://sciencedirect.com/science/article/pii/ S0828282X21006243
- Myocarditis, pericarditis and cardiomyopathy after COVID-19 vaccination: https://sciencedirect.com/science/article/pii/S1443950621011562
- Myocarditis and pericarditis after COVID-19 vaccination: https://jamanetwork.com/journals/jama/fullarticle/2782900
- Pericarditis after administration of BNT162b2 mRNA COVID-19 mRNA vaccine:https://sciencedirect.com/science/article/pii/S1885585721002218
- Epidemiology and clinical features of myocarditis/pericarditis before the introduction of COVID-19 mRNAvaccine in Korean children: a multicenter study https:// bvsalud.org/global-literature-on-novel-coronavirus-2019-ncov/resourc e/en/ covidwho-1360706.
- Myocarditis, pericarditis, and cardiomyopathy after COVID-19 vaccination: https://ncbi.nlm.nih.gov/34340927/
- Clinical Guidance for Young People with Myocarditis and Pericarditis after Vaccination with COVID-19mRNA: https://cps.ca/en/documents/position/ clinical-guidance-for-youth-with-myocarditis-and-pericarditis
- Myocarditis / pericarditis associated with COVID-19 vaccine: https://science.gc.ca/eic/site/063.nsf/eng/h_98291.html
- Acute myocarditis after the second dose of SARS-CoV-2 vaccine: serendipity or causal relationship: https://pubmed.ncbi.nlm.nih.gov/34236331/
- Pericarditis after administration of COVID-19 mRNA BNT162b2 vaccine: https://ncbi.nlm.nih.gov/34364831/
- Unusual presentation of acute pericarditis after vaccination against SARS-COV-2 mRNA-1237 Modern: https://pubmed.ncbi.nlm.nih.gov/34447639/
- A case series of acute pericarditis after vaccination with COVID-19 in the context of recent reports fromEurope and the United States: https://pubmed.ncbi.nlm.nih.gov/ 34635376/
- Acute pericarditis and cardiac tamponade after vaccination with Covid-19: https://ncbi.nlm.nih.gov/34749492/
- Pericarditis after administration of the BNT162b2 mRNA vaccine COVID-19: https://ncbi.nlm.nih.gov/34149145/
- Case report: symptomatic pericarditis post COVID-19 vaccination: https://ncbi.nlm.nih.gov/34693198/.
- Acute myelitis and ChAdOx1 nCoV-19 vaccine: coincidental or causal association: https://sciencedirect.com/science/article/pii/S0165572821002137
- Acute transverse myelitis (ATM): clinical review of 43 patients with COVID-19- associated ATM and 3 serious adverse events of post-vaccination ATM with ChAdOx1 nCoV-19 vaccine (AZD1222):https://pubmed.ncbi.nlm.nih.gov/33981305/
- Transverse myelitis induced by SARS-CoV-2 vaccination: https://ncbi.nlm.nih.gov/34458035/
- Acute transverse myelitis (ATM): clinical review of 43 patients with COVID-19- associated ATM and 3 serious adverse events of post-vaccination ATM with ChAdOx1 nCoV-19 (AZD1222) vaccine:https://pubmed.ncbi.nlm.nih.gov/33981305/.
- Acute transverse myelitis after COVID-19 vaccination: https://ncbi.nlm.nih.gov/34684047/.
- Extensive longitudinal transverse myelitis after ChAdOx1 nCOV-19 vaccine: case report: https://pubmed.ncbi.nlm.nih.gov/34641797/.
- Acute transverse myelitis after SARS-CoV-2 vaccination: case report and review of the literature: https://pubmed.ncbi.nlm.nih.gov/34482455/.
- Acute transverse myelitis following inactivated COVID-19 vaccine: https://ncbi.nlm.nih.gov/34370410/
- Acute transverse myelitis after COVID-19 vaccination: https://ncbi.nlm.nih.gov/34579245/.
- A case of longitudinally extensive transverse myelitis following Covid-19 vaccination: https://pubmed.ncbi.nlm.nih.gov/34182207/
- Post COVID-19 transverse myelitis; a case report with review of the literature: https://ncbi.nlm.nih.gov/34457267/.
- Acute bilateral bilateral optic neuritis/chiasm with longitudinal extensive transverse myelitis in long-standing stable multiple sclerosis after vector-based vaccination against SARS-CoV-2: https://pubmed.ncbi.nlm.nih.gov/34131771/
- Extensive longitudinal transverse myelitis following AstraZeneca COVID-19 vaccination: https://pubmed.ncbi.nlm.nih.gov/34507942/.
- Extensive longitudinal transverse myelitis following AstraZeneca COVID-19 vaccination: https://pubmed.ncbi.nlm.nih.gov/34507942/.
- Longitudinally extensive cervical myelitis after vaccination with inactivated virus based COVID-19 vaccine: https://pubmed.ncbi.nlm.nih.gov/34849183/
- Perimyocarditis in adolescents after Pfizer-BioNTech COVID-19 vaccine: https://oup.com/jpids/advance-article/doi/10.1093/jpids/piab060/6329543
- Perimyocarditis in adolescents after Pfizer-BioNTech COVID-19 vaccine: https://ncbi.nlm.nih.gov/34319393/
- Unusual presentation of acute perimyocarditis after modern SARS-COV-2 mRNA-1237 vaccination: https://pubmed.ncbi.nlm.nih.gov/34447639/
- Perimyocarditis after the first dose of mRNA-1273 SARS-CoV-2 (Modern) mRNA-1273 vaccine in a young healthy male: case report: https://biomedcentral.com/articles/10.1186/s12872-021-02183
- Acute perimyocarditis after the first dose of COVID-19 mRNA vaccine: https://ncbi.nlm.nih.gov/34515024/
- Perimyocarditis after vaccination with COVID-19: https://pubmed.ncbi.nlm.nih.gov/ 34866957/
- Tinoco, , Leite, S., Faria, B., Cardoso, S., Von Hafe, P., Dias, G., . . . Lourenco, A. (2021). PerimyocarditisFollowing COVID-19 Vaccination. Clin Med Insights Cardiol, 15, 11795468211056634. doi:10.1177/11795468211056634. https://
www.ncbi.nlm.nih.gov/pubmed/34866957
- Jhaveri, , Adler-Shohet, F. C., Blyth, C. C., Chiotos, K., Gerber, J. S., Green,
M., . . . Zaoutis, T. (2021). Weighing the Risks of Perimyocarditis With the Benefits of SARS-CoV-2 mRNAVaccination in Adolescents. J Pediatric Infect Dis Soc, 10(10), 937-939. doi:10.1093/jpids/piab061.https://www.ncbi.nlm.nih.gov/pubmed/34270752
- Khogali, F., & Abdelrahman, R. (2021). Unusual Presentation of Acute Perimyocarditis Following SARS-COV-2 mRNA-1237 Moderna Cureus, 13(7), e16590. doi:10.7759/cureus.16590. https://www.ncbi.nlm.nih.gov/pubmed/ 34447639
- Hasnie, A., Hasnie, U. A., Patel, N., Aziz, M. U., Xie, M., Lloyd, S. G., & Prabhu, S.
- (2021). Perimyocarditis following first dose of the mRNA-1273 SARS-CoV-2 (Moderna) vaccine in ahealthy young male: a case report. BMC Cardiovasc Disord, 21(1), 375. doi:10.1186/s12872-021-02183-3.https://www.ncbi.nlm.nih.gov/pubmed/
34348657
- Intracerebral haemorrhage due to thrombosis with thrombocytopenia syndrome after COVID-19 vaccination:the first fatal case in Korea: https://pubmed.ncbi.nlm.nih.gov/ 34402235/
- Intracerebral haemorrhage twelve days after vaccination with ChAdOx1 nCoV-19: https://pubmed.ncbi.nlm.nih.gov/34477089/
- Neurosurgical considerations regarding decompressive craniectomy for intracerebral hemorrhage after SARS-CoV-2 vaccination in vaccine-induced thrombotic thrombocytopenia-VITT: https://pubmed.ncbi.nlm.nih.gov/34202817/
- First dose of ChAdOx1 and BNT162b2 COVID-19 vaccines and thrombocytopenic, thromboembolic, and hemorrhagic events in Scotland: https:// ncbi.nlm.nih.gov/34108714/
- Large hemorrhagic stroke after vaccination against ChAdOx1 nCoV-19: a case report: https://pubmed.ncbi.nlm.nih.gov/34273119/
- Major hemorrhagic stroke after ChAdOx1 nCoV-19 vaccination: a case report: https://ncbi.nlm.nih.gov/34273119/
- Aphasia seven days after the second dose of an mRNA-based SARS-CoV-2 Brain MRI revealed an intracerebral haemorrhage (ICBH) in the left temporal lobe in a 52-year-old man. https://www.sciencedirect.com/science/article/pii/ S2589238X21000292#f0005
- Incidence of acute ischemic stroke after coronavirus vaccination in Indonesia: case series: https://pubmed.ncbi.nlm.nih.gov/34579636/
- Autoimmune hepatitis developing after coronavirus disease vaccine 2019 (COVID-19): causality orvictim?: https://pubmed.ncbi.nlm.nih.gov/33862041/
- Autoimmune hepatitis triggered by vaccination against SARS-CoV-2: https://ncbi.nlm.nih.gov/34332438/
- Acute autoimmune-like hepatitis with atypical antimitochondrial antibody after vaccination with COVID-19 mRNA: a new clinical entity: https:// ncbi.nlm.nih.gov/34293683/.
- Autoimmune hepatitis after COVID vaccine: https://pubmed.ncbi.nlm.nih.gov/ 34225251/
- Hepatitis C virus reactivation after COVID-19 vaccination: a case report: https://ncbi.nlm.nih.gov/34512037/
- Autoimmune hepatitis developing after ChAdOx1 nCoV-19 vaccine (Oxford- AstraZeneca): https://pubmed.ncbi.nlm.nih.gov/34171435/
- Autoimmune hepatitis triggered by SARS-CoV-2 vaccination: https://ncbi.nlm.nih.gov/34332438/.
- Immune-mediated hepatitis with the Moderna vaccine is no longer a coincidence but confirmed: https://sciencedirect.com/science/article/pii/S0168827821020936
- Facial nerve palsy following administration of COVID-19 mRNA vaccines: analysis of self-report database: https://sciencedirect.com/science/article/pii/ S1201971221007049
- COVID-19 vaccination association and facial nerve palsy: A case-control study: https://pubmed.ncbi.nlm.nih.gov/34165512/
- Sequential contralateral facial nerve palsy after first and second doses of COVID-19 vaccine: https://pubmed.ncbi.nlm.nih.gov/34281950/.
- Peripheral facial nerve palsy after vaccination with BNT162b2 (COVID-19): https://ncbi.nlm.nih.gov/33734623/
- Facial nerve palsy after administration of COVID-19 mRNA vaccines: analysis of self- report database: https://pubmed.ncbi.nlm.nih.gov/34492394/
- A case of acute demyelinating polyradiculoneuropathy with bilateral facial palsy following ChAdOx1 nCoV-19vaccination: https://pubmed.ncbi.nlm.nih.gov/34272622/
- Neurological symptoms and neuroimaging alterations related to COVID-19 vaccine: cause or coincidence: https://sciencedirect.com/science/article/pii/ S0899707121003557.
- Neurological symptoms and neuroimaging alterations related to COVID-19 vaccine: cause or coincidence?: https://pubmed.ncbi.nlm.nih.gov/34507266/
- Spectrum of neurological complications after COVID-19 vaccination: https://ncbi.nlm.nih.gov/34719776/.
- n-hospital observational study of neurological disorders in patients recently vaccinated with COVID-19 mRNA vaccines: https://pubmed.ncbi.nlm.nih.gov/ 34688190/
- Neurological side effects of SARS-CoV-2 vaccines: https://pubmed.ncbi.nlm.nih.gov/ 34750810/
- Neurological complications after the first dose of COVID-19 vaccines and SARS- CoV-2 infection: https://pubmed.ncbi.nlm.nih.gov/34697502/
- Lobar hemorrhage with ventricular rupture shortly after the first dose of an mRNA- based SARS-CoV-2 vaccine: https://ncbi.nlm.nih.gov/labs/pmc/articles/ PMC8553377/
- Retinal hemorrhage after SARS-CoV-2 vaccination: https://pubmed.ncbi.nlm.nih.gov/ 34884407/.
- Lobar hemorrhage with ventricular rupture shortly after the first dose of a SARS- CoV-2 mRNA-based SARS-CoV-2 vaccine: https://pubmed.ncbi.nlm.nih.gov/ 34729467/
- Acral hemorrhage after administration of the second dose of SARS-CoV-2 A post-vaccination reaction: https://pubmed.ncbi.nlm.nih.gov/34092400/742.
- Fatal cerebral hemorrhage after COVID-19 vaccine: https://pubmed.ncbi.nlm.nih.gov/ 33928772/
- Intracerebral hemorrhage associated with vaccine-induced thrombotic thrombocytopenia after ChAdOx1nCOVID-19 vaccination in a pregnant woman: https://pubmed.ncbi.nlm.nih.gov/34261297/
- Immune-mediated disease outbreaks or recent-onset disease in 27 subjects after mRNA/DNA vaccination against SARS-CoV-2: https://pubmed.ncbi.nlm.nih.gov/ 33946748/
- Severe autoimmune hemolytic autoimmune anemia after receiving SARS-CoV-2 mRNA vaccine: https://pubmed.ncbi.nlm.nih.gov/34549821/
- Severe autoimmune hemolytic anemia after receipt of SARS-CoV-2 mRNA vaccine:https://onlinelibrary.wiley.com/doi/10.1111/trf.16672
- https://ncbi.nlm.nih.gov/pubmed/34127481
- Autoimmune encephalitis after ChAdOx1-S SARS-CoV-2 vaccination: https://ncbi.nlm.nih.gov/34846583/
- Immune-mediated disease outbreaks or new-onset disease in 27 subjects after mRNA/DNAvaccination against SARS-CoV-2: https://pubmed.ncbi.nlm.nih.gov/ 33946748/
- Myocarditis, pericarditis and cardiomyopathy after COVID-19 vaccination: https://sciencedirect.com/science/article/pii/S1443950621011562
- Takotsubo cardiomyopathy after vaccination with mRNA COVID-19: https://sciencedirect.com/science/article/pii/S1443950621011331
- Takotsubo (stress) cardiomyopathy after vaccination with ChAdOx1 nCoV-19: https://ncbi.nlm.nih.gov/34625447/
- Takotsubo cardiomyopathy after coronavirus 2019 vaccination in patient on maintenance hemodialysis: https://pubmed.ncbi.nlm.nih.gov/34731486/.
- Takotsubo syndrome after COVID-19 vaccination: https://pubmed.ncbi.nlm.nih.gov/ 34539938/.
- Transient cardiac injury in adolescents receiving the BNT162b2 mRNA COVID-19 vaccine: https://journals.lww.com/pidj/Abstract/9000/ Transient_Cardiac_Injury_in_Adolesce nts_Receiving.95800.aspx
- Snapiri, O., Rosenberg Danziger, C., Shirman, N., Weissbach, , Lowenthal, A., Ayalon, I., . . . Bilavsky, E.(2021). Transient Cardiac Injury in Adolescents Receiving the BNT162b2 mRNA COVID-19 Vaccine. Pediatr Infect Dis J, 40(10), e360-e363. doi:10.1097/INF.0000000000003235. https://www.ncbi.nlm.nih.gov/pubmed/ 34077949
- Fazlollahi, , Zahmatyar, M., Noori, M., Nejadghaderi, S. A., Sullman, M. J. M., Shekarriz-Foumani, R., . . . Safiri, S. (2021). Cardiac complications following mRNA COVID-19 vaccines: A systematic review of case reports and case series. Rev Med Virol, e2318. doi:10.1002/rmv.2318.https://www.ncbi.nlm.nih.gov/pubmed/34921468
- Ho, S., Sia, C. H., Ngiam, J. N., Loh, P. H., Chew, N. W., Kong, W. K., & Poh, K. K.
(2021). A review of COVID-19 vaccination and the reported cardiac manifestations. Singapore Med J. doi:10.11622/smedj.2021210. https://www.ncbi.nlm.nih.gov/ pubmed/34808708
- Temporal relationship between the second dose of BNT162b2 mRNA Covid-19 vaccine and cardiacinvolvement in a patient with previous SARS-COV-2 infection: https://sciencedirect.com/science/article/pii/S2352906721000622
- Sessa, F., Salerno, M., Esposito, M., Di Nunno, N., Zamboni, P., & Pomara, C. (2021). Autopsy Findingsand Causality Relationship between Death and COVID-19 Vaccination: A Systematic Review. J Clin Med, 10(24). doi:10.3390/
jcm10245876. https://www.ncbi.nlm.nih.gov/pubmed/34945172
- Post-mortem investigation of deaths after vaccination with COVID-19 vaccines: https://pubmed.ncbi.nlm.nih.gov/34591186/
- A look at the role of postmortem immunohistochemistry in understanding the inflammatory pathophysiologyof COVID-19 disease and vaccine-related thrombotic adverse events: a narrative review: https://pubmed.ncbi.nlm.nih.gov/34769454/
- COVID-19 vaccine and death: causality algorithm according to the WHO eligibility diagnosis: https://pubmed.ncbi.nlm.nih.gov/34073536/
- Post-mortem investigation of deaths after vaccination with COVID-19 vaccines: https://pubmed.ncbi.nlm.nih.gov/34591186/
- Rhabdomyolysis and fasciitis induced by the COVID-19 mRNA vaccine: https://ncbi.nlm.nih.gov/34435250/
- COVID-19 vaccine-induced rhabdomyolysis: case report with literature review: https://pubmed.ncbi.nlm.nih.gov/34186348/
- COVID-19 vaccine-induced rhabdomyolysis: case report with review of the literature: https://sciencedirect.com/science/article/pii/S1871402121001880
- Rhabdomyolysis and fasciitis induced by COVID-19 mRNA vaccine: https://ncbi.nlm.nih.gov/34435250/.
- Case report: ANCA-associated vasculitis presenting with rhabdomyolysis and crescentic Pauci-Inmuneglomerulonephritis after vaccination with Pfizer-BioNTech COVID-19 mRNA: https://pubmed.ncbi.nlm.nih.gov/34659268/
- Thrombotic thrombocytopenic purpura after vaccination with COV2-S: https://pubmed.ncbi.nlm.nih.gov/33980419/
- Thrombotic thrombocytopenic purpura: a new threat after COVID bnt162b2 vaccine: https://pubmed.ncbi.nlm.nih.gov/34264514/.
- Severe immune thrombocytopenic purpura after SARS-CoV-2 vaccine: https://ncbi.nlm.nih.gov/34754937/
- Immune thrombocytopenic purpura associated with COVID-19 mRNA vaccine Pfizer- BioNTech BNT16B2b2: https://pubmed.ncbi.nlm.nih.gov/34077572/
- Myocarditis and other cardiovascular complications of COVID-19 mRNA-based COVID-19 vaccineshttps://cureus.com/articles/61030-myocarditis-and-other- cardiovascular-complications-of-the-mrna-based-covid-19-vaccines
- Cardiovascular magnetic resonance imaging findings in young adult patients with acute myocarditis after COVID-19 mRNA vaccination: a case series: https://jcmr-biomedcentral.com/articles/10.1186/s12968-021-00795-4
- Be alert to the risk of adverse cardiovascular events after COVID-19 vaccination: https://xiahepublishing.com/m/2472-0712/ERHM-2021-00033
- Myocarditis and other cardiovascular complications of mRNA-based COVID-19 vaccines: https://pubmed.ncbi.nlm.nih.gov/34277198/
- Acute hyperactive encephalopathy following COVID-19 vaccination with dramatic response to methylprednisolone: a case report: https://sciencedirect.com/science/article/pii/S2049080121007536
- Post-vaccinal encephalitis after ChAdOx1 nCov-19: https://pubmed.ncbi.nlm.nih.gov/ 34324214/
- Acute disseminated encephalomyelitis following vaccination against SARS-CoV-2: https://pubmed.ncbi.nlm.nih.gov/34325334/
- Acute hyperactive encephalopathy following COVID-19 vaccination with dramatic response to methylprednisolone: case report: https://pubmed.ncbi.nlm.nih.gov/ 34512961/
- Minimal change disease with severe acute kidney injury after Oxford-AstraZeneca COVID-19 vaccine: case report: https://pubmed.ncbi.nlm.nih.gov/34242687/.
- Acute kidney injury with macroscopic hematuria and IgA nephropathy after COVID-19 vaccination: https://pubmed.ncbi.nlm.nih.gov/34352309/
- AstraZeneca): https://pubmed.ncbi.nlm.nih.gov/34362727/
- Minimal change disease and acute kidney injury after Pfizer-BioNTech COVID-19 vaccine: https://pubmed.ncbi.nlm.nih.gov/34000278/
- Severe relapse of multiple sclerosis after COVID-19 vaccination: a case report:https://pubmed.ncbi.nlm.nih.gov/34447349/
- Acute relapse and impaired immunization after COVID-19 vaccination in a patient with multiple sclerosis treated with rituximab: https://pubmed.ncbi.nlm.nih.gov/ 34015240
- Humoral response induced by Prime-Boost vaccination with ChAdOx1 nCoV-19 and BNT162b2 mRNAvaccines in a patient with multiple sclerosis treated with teriflunomide: https://pubmed.ncbi.nlm.nih.gov/34696248/
- A rare case of Henoch-Schönlein purpura after a case report of COVID-19 vaccine: https://pubmed.ncbi.nlm.nih.gov/34518812/
- Henoch-Schönlein purpura occurring after vaccination with COVID-19: https://ncbi.nlm.nih.gov/34247902/.
- Henoch-Schönlein purpura following the first dose of COVID-19 viral vector vaccine: case report: https://pubmed.ncbi.nlm.nih.gov/34696186/.
- Blood clots and bleeding episodes after BNT162b2 and ChAdOx1 nCoV-19 vaccination: analysis ofEuropean data: https://sciencedirect.com/science/ article/pii/S0896841121000937
- Association between ChAdOx1 nCoV-19 vaccination and bleeding episodes: large population-based cohort study: https://pubmed.ncbi.nlm.nih.gov/34479760/.
- Association between ChAdOx1 nCoV-19 vaccination and bleeding episodes: large population-based cohort study: https://pubmed.ncbi.nlm.nih.gov/34479760/.
- Cutaneous adverse effects of available COVID-19 vaccines: https://ncbi.nlm.nih.gov/34518015/
- Rare cutaneous adverse effects of COVID-19 vaccines: a case series and review of the literature: https://pubmed.ncbi.nlm.nih.gov/34363637/
- Cutaneous adverse reactions of 35,229 doses of COVID-19 Sinovac and AstraZeneca vaccine COVID-19:a prospective cohort study in health care workers: https://pubmed.ncbi.nlm.nih.gov/34661934/
- A case series of skin reactions to COVID-19 vaccine in the Department of Dermatology at Loma LindaUniversity: https://pubmed.ncbi.nlm.nih.gov/34423106/
- Skin reactions reported after Moderna and Pfizer’s COVID-19 vaccination: a study based on a registry of 414 cases: https://pubmed.ncbi.nlm.nih.gov/33838206/
- Skin reactions after vaccination against SARS-COV-2: a nationwide Spanish cross- sectional study of 405 cases: https://pubmed.ncbi.nlm.nih.gov/34254291/
- Coagulopathies after SARS-CoV-2 vaccination may derive from a combined effect of SARS-CoV-2 spike protein and adenovirus vector-activated signaling pathways: https://pubmed.ncbi.nlm.nih.gov/34639132/
- Diffuse prothrombotic syndrome after administration of ChAdOx1 nCoV-19 vaccine: case report:https://pubmed.ncbi.nlm.nih.gov/34615534/
- Calcaterra, G., Bassareo, P. P., Barilla, F., Romeo, F., & Mehta, J. L. (2022). Concerning the unexpectedprothrombotic state following some coronavirus disease 2019 vaccines. J Cardiovasc Med (Hagerstown), 23(2), 71-74. doi:10.2459/JCM.
- https://www.ncbi.nlm.nih.gov/pubmed/34366403
- Post-vaccination multisystem inflammatory syndrome in adults without evidence of prior SARS-CoV-2 infection: https://pubmed.ncbi.nlm.nih.gov/34852213/
- Buchhorn, R., Meyer, C., Schulze-Forster, K., Junker, J., & Heidecke, H. (2021). Autoantibody Release in Children after Corona Virus mRNA Vaccination: A Risk Factor of Multisystem Inflammatory Syndrome?Vaccines (Basel), 9(11). doi:10.3390/ vaccines9111353. https://ncbi.nlm.nih.gov/pubmed/34835284
- Chai, Q., Nygaard, U., Schmidt, R. C., Zaremba, T., Moller, M., & Thorvig, C. M. (2022). Multisystem inflammatory syndrome in a male adolescent after his second Pfizer-BioNTech COVID-19 vaccine. Acta Paediatr, 111(1), 125-127. doi:10.1111/apa. 16141.
- Vogt-Koyanagi-Harada syndrome after COVID-19 and ChAdOx1 nCoV-19 (AZD1222) vaccination: https://pubmed.ncbi.nlm.nih.gov/34462013/.
- Reactivation of Vogt-Koyanagi-Harada disease under control for more than 6 years, after anti-SARS-CoV-2 vaccination: https://pubmed.ncbi.nlm.nih.gov/34224024/
- Fatal systemic capillary leak syndrome after SARS-COV-2 vaccination in a patient with multiple myeloma: https://pubmed.ncbi.nlm.nih.gov/34459725/
- Systemic capillary extravasation syndrome following vaccination with ChAdOx1 nCOV-19 (Oxford-AstraZeneca): https://pubmed.ncbi.nlm.nih.gov/34362727/
- Induction and exacerbation of subacute cutaneous lupus erythematosus erythematosus after mRNA- oradenoviral vector-based SARS-CoV-2 vaccination: https://pubmed.ncbi.nlm.nih.gov/34291477/
- Ntouros, P. , Vlachogiannis, N. I., Pappa, M., Nezos, A., Mavragani, C. P., Tektonidou, M. G., . . . Sfikakis,P. P. (2021). Effective DNA damage response after acute but not chronic immune challenge: SARS-CoV-2 vaccine versus Systemic Lupus Erythematosus. Clin Immunol, 229, 108765.doi:10.1016/j.clim.2021.108765. https://www.ncbi.nlm.nih.gov/pubmed/34089859
- Petechiae and peeling of fingers after immunization with BTN162b2 messenger RNA (mRNA)-based COVID-19 vaccine: https://pubmed.ncbi.nlm.nih.gov/34513435/
- Petechial rash associated with CoronaVac vaccination: first report of cutaneous side effects before phase 3 results: https://ejhp.bmj.com/content/early/2021/05/23/ ejhpharm-2021-002794
- Purpuric rash and thrombocytopenia after mRNA-1273 (Modern) COVID-19 vaccine: https://ncbi.nlm.nih.gov/pmc/articles/PMC7996471/
- Generalized purpura annularis telangiectodes after SARS-CoV-2 mRNA vaccination:https://pubmed.ncbi.nlm.nih.gov/34236717/
- Pulmonary embolism, transient ischemic attack, and thrombocytopenia after Johnson & Johnson COVID-19 vaccine: https://pubmed.ncbi.nlm.nih.gov/34261635/
- A case of acute pulmonary embolism after immunization with SARS-CoV-2 mRNA: https://pubmed.ncbi.nlm.nih.gov/34452028/
- Onset / outbreak of psoriasis after Corona virus ChAdOx1 nCoV-19 vaccine (Oxford- AstraZeneca / Covishield): report of two cases: https://pubmed.ncbi.nlm.nih.gov/ 34350668/
- Exacerbation of plaque psoriasis after COVID-19 inactivated mRNA and BNT162b2 vaccines: report of two cases: https://pubmed.ncbi.nlm.nih.gov/34427024/
- Miller Fisher syndrome after Pfizer COVID-19 vaccine: https://ncbi.nlm.nih.gov/34817727/.
- Miller Fisher syndrome after 2019 BNT162b2 mRNA coronavirus vaccination: https://ncbi.nlm.nih.gov/34789193/.
- Nephrotic syndrome after ChAdOx1 nCoV-19 vaccine against SARScoV-2: https://ncbi.nlm.nih.gov/34250318/.
- New-onset nephrotic syndrome after Janssen COVID-19 vaccination: case report and literature review: https://pubmed.ncbi.nlm.nih.gov/34342187/
- Hematuria, a generalized petechial rash and headaches after Oxford AstraZeneca ChAdOx1 nCoV-19 vaccination: https://pubmed.ncbi.nlm.nih.gov/34620638/
- A case of outbreak of macroscopic hematuria and IgA nephropathy after SARS- CoV-2 vaccination: https://pubmed.ncbi.nlm.nih.gov/33932458/
- Bullous drug eruption after the second dose of COVID-19 mRNA-1273 (Moderna) vaccine: Case report: https://sciencedirect.com/science/article/pii/ S1876034121001878.
- Widespread fixed bullous drug eruption after vaccination with ChAdOx1 nCoV-19: https://pubmed.ncbi.nlm.nih.gov/34482558/
- Hemophagocytic lymphohistiocytosis after vaccination with ChAdOx1 nCov-19: https://pubmed.ncbi.nlm.nih.gov/34406660/.
- Hemophagocytic lymphohistiocytosis following COVID-19 vaccination (ChAdOx1 nCoV-19): https://pubmed.ncbi.nlm.nih.gov/34862234/
- Isolated pulmonary embolism after COVID vaccination: 2 case reports and a review of acute pulmonary embolism complications and follow-up: https:// ncbi.nlm.nih.gov/34804412/
- Myocardial infarction, stroke, and pulmonary embolism after BNT162b2 mRNA COVID-19 vaccine in persons aged 75 years or older: https:// ncbi.nlm.nih.gov/34807248/
- Beware of neuromyelitis optica spectrum disorder after vaccination with inactivated virus for COVID-19: https://pubmed.ncbi.nlm.nih.gov/34189662/
- Neuromyelitis optica in a healthy woman after vaccination against severe acute respiratory syndromecoronavirus 2 mRNA-1273: https://pubmed.ncbi.nlm.nih.gov/ 34660149/
- Shingles-like skin lesion after vaccination with AstraZeneca for COVID-19: a case report: https://pubmed.ncbi.nlm.nih.gov/34631069/
- Recurrent herpes zoster after COVID-19 vaccination in patients with chronic urticaria on cyclosporine treatment – A report of 3 cases: https://pubmed.ncbi.nlm.nih.gov/ 34510694/
- Blood clots and bleeding after BNT162b2 and ChAdOx1 nCoV-19 vaccination: an analysis of European data: https://pubmed.ncbi.nlm.nih.gov/34174723/
- Antiphospholipid antibodies and risk of thrombophilia after COVID-19 vaccination: the straw that breaks the camel’s back?: https://docs.google.com/document/d/ 1XzajasO8VMMnC3CdxSBKks1o7kiOLXFQ
- First report of a de novo iTTP episode associated with a COVID-19 mRNA-based anti-COVID-19 vaccine: https://pubmed.ncbi.nlm.nih.gov/34105244/
- New-onset refractory status epilepticus after chadox1 ncov-19 vaccination: https://sciencedirect.com/science/article/pii/s0165572821001569
- Acute-onset central serous retinopathy after immunization with COVID-19 mRNA vaccine:.https://sciencedirect.com/science/article/pii/S2451993621001456.
- Late cutaneous reactions after administration of COVID-19 mRNA vaccines: https://sciencedirect.com/science/article/pii/S2213219821007996
- COVID-19 RNA-based vaccines and the risk of prion disease: https://com/pdfs/covid19rna-based-vaccines-and-the-risk-of-prion-dis ease-1503.pdf
- This study notes that 115 pregnant women lost their babies, out of 827 who participated in a study on thesafety of covid-19 vaccines: https://nejm.org/doi/ full/10.1056/NEJMoa2104983.
- Process-related impurities in the ChAdOx1 nCov-19 vaccine: https://researchsquare.com/article/rs-477964/v1
- COVID-19 mRNA vaccine causing CNS inflammation: a case series: https://springer.com/article/10.1007/s00415-021-10780-7
- A systematic review of cases of CNS demyelination following COVID-19 vaccination: https://pubmed.ncbi.nlm.nih.gov/34839149/
- Reported orofacial adverse effects from COVID-19 vaccines: the known and the unknown: https://pubmed.ncbi.nlm.nih.gov/33527524/
- Fatal brain haemorrhage after COVID-19 vaccine: https://pubmed.ncbi.nlm.nih.gov/ 33928772/
- Acute retinal necrosis due to varicella zoster virus reactivation after vaccination with BNT162b2 COVID-19 mRNA: https://pubmed.ncbi.nlm.nih.gov/34851795/.
- Nerve and muscle adverse events after vaccination with COVID-19: a systematic review and meta-analysis of clinical trials: https://pubmed.ncbi.nlm.nih.gov/ 34452064/.
- Transient oculomotor paralysis after administration of RNA-1273 messenger vaccine for SARS-CoV-2 diplopia after COVID-19 vaccine: https://pubmed.ncbi.nlm.nih.gov/ 34369471/
- Parsonage-Turner syndrome associated with SARS-CoV-2 or SARS-CoV-2 vaccination. Comment on:“Neuralgic amyotrophy and COVID-19 infection: 2 cases of accessory spinal nerve palsy” by Coll et al.Articular Spine 2021; 88: 10519: https://pubmed.ncbi.nlm.nih.gov/34139321/.
- Bilateral acute macular neuroretinopathy after SARS-CoV-2 vaccination: https://ncbi.nlm.nih.gov/34287612/
- Lipschütz ulcers after AstraZeneca COVID-19 vaccination: https://ncbi.nlm.nih.gov/34366434/.
- Amyotrophic Neuralgia secondary to Vaxzevri vaccine (AstraZeneca) COVID-19: https://pubmed.ncbi.nlm.nih.gov/34330677/
- Polyarthralgia and myalgia syndrome after vaccination with ChAdOx1 nCOV-19: https://pubmed.ncbi.nlm.nih.gov/34463066/
- Three cases of subacute thyroiditis after SARS-CoV-2 vaccination: post-vaccination ASIA syndrome: https://pubmed.ncbi.nlm.nih.gov/34043800/.
- Bilateral immune-mediated keratolysis after immunization with SARS-CoV-2 recombinant viral vectorvaccine: https://pubmed.ncbi.nlm.nih.gov/34483273/.
- Reactive arthritis after COVID-19 vaccination: https://pubmed.ncbi.nlm.nih.gov/ 34033732/.
- Thymic hyperplasia after Covid-19 mRNA-based vaccination with Covid-19: https://ncbi.nlm.nih.gov/34462647/
- Tolosa-Hunt syndrome occurring after COVID-19 vaccination: https://ncbi.nlm.nih.gov/34513398/
- Hailey-Hailey disease exacerbation after SARS-CoV-2 vaccination: https://ncbi.nlm.nih.gov/34436620/
- Rituximab-induced acute lympholysis and pancytopenia following vaccination with COVID-19: https://pubmed.ncbi.nlm.nih.gov/34429981/
- Vaccine-induced interstitial lung disease: a rare reaction to COVID-19 vaccine: https://pubmed.ncbi.nlm.nih.gov/34510014/.
- Vesiculobullous cutaneous reactions induced by COVID-19 mRNA vaccine: report of four cases and review of the literature: https://pubmed.ncbi.nlm.nih.gov/34236711/
- Collection of complement-mediated and autoimmune-mediated hematologic conditions after SARS-CoV-2vaccination: https://ashpublications.org/bloodadvances/ article/5/13/2794/476324/Autoimmune-and-complement-mediated-hematologic
- COVID-19 vaccines induce severe hemolysis in paroxysmal nocturnal hemoglobinuria:https://ashpublications.org/blood/article/137/26/3670/475905/ COVID-19-vaccines-induce-severe-hemolysis-in
- Headache attributed to COVID-19 (SARS-CoV-2 coronavirus) vaccination with the ChAdOx1 nCoV-19 (AZD1222) vaccine: a multicenter observational cohort study: https://pubmed.ncbi.nlm.nih.gov/34313952/
- Mrna COVID vaccines dramatically increase endothelial inflammatory markers and risk of Acute CoronarySyndrome as measured by PULS cardiac testing: a caution:https://ahajournals.org/doi/10.1161/circ.144.suppl_1.10712
- ANCA glomerulonephritis following Modern COVID-19 vaccination: https://ncbi.nlm.nih.gov/34081948/
- Unique imaging findings of neurologic phantosmia after Pfizer-BioNtech COVID-19 vaccination: a case report: https://pubmed.ncbi.nlm.nih.gov/34096896/
- Bilateral uveitis after inoculation with COVID-19 vaccine: a case report: https://sciencedirect.com/science/article/pii/S1201971221007797
- Extensive investigations revealed consistent pathophysiologic alterations after vaccination with COVID-19 vaccines: https://nature.com/articles/
s41421-021-00329-3
- Gross hematuria after severe acute respiratory syndrome coronavirus 2 vaccination in 2 patients with IgA nephropathy: https://pubmed.ncbi.nlm.nih.gov/33771584/
- Inflammatory myositis after vaccination with ChAdOx1: https://ncbi.nlm.nih.gov/34585145/
- An outbreak of Still’s disease after COVID-19 vaccination in a 34-year-old patient: https://pubmed.ncbi.nlm.nih.gov/34797392/
- Case report: Pityriasis rosea-like rash after vaccination with COVID-19: https://ncbi.nlm.nih.gov/34557507/
- Acute eosinophilic pneumonia associated with anti-COVID-19 vaccine AZD1222: https://pubmed.ncbi.nlm.nih.gov/34812326/.
- Sweet’s syndrome after Oxford-AstraZeneca COVID-19 vaccine (AZD1222) in an elderly woman: https://pubmed.ncbi.nlm.nih.gov/34590397/
- Sudden sensorineural hearing loss after COVID-19 vaccination: https://ncbi.nlm.nih.gov/34670143/.
- Prevalence of serious adverse events among health care professionals after receiving the first dose ofChAdOx1 nCoV-19 coronavirus vaccine (Covishield) in Togo, March 2021: https://pubmed.ncbi.nlm.nih.gov/34819146/.
- A case of toxic epidermal necrolysis after vaccination with ChAdOx1 nCoV-19 (AZD1222): https://pubmed.ncbi.nlm.nih.gov/34751429/.
- Ocular adverse events following COVID-19 vaccination: https://ncbi.nlm.nih.gov/34559576/
- Depression after ChAdOx1-S / nCoV-19 vaccination: https://ncbi.nlm.nih.gov/34608345/.
- Pancreas allograft rejection after ChAdOx1 nCoV-19 vaccine: https://ncbi.nlm.nih.gov/34781027/
- Acute hemichorea-hemibalismus after COVID-19 (AZD1222) vaccination: https://ncbi.nlm.nih.gov/34581453/
- Recurrence of alopecia areata after covid-19 vaccination: a report of three cases in Italy: https://pubmed.ncbi.nlm.nih.gov/34741583/
- Two cases of Graves’ disease after SARS-CoV-2 vaccination: an autoimmune / inflammatory syndromeinduced by adjuvants: https://pubmed.ncbi.nlm.nih.gov/ 33858208/
- Cardiovascular, neurological, and pulmonary events after vaccination with BNT162b2, ChAdOx1nCoV-19, and COV2.S vaccines: an analysis of European data: https://pubmed.ncbi.nlm.nih.gov/34710832/
- Change in blood viscosity after COVID-19 vaccination: estimation for persons with underlying metabolic syndrome: https://pubmed.ncbi.nlm.nih.gov/34868465/
- Eosinophilic dermatosis after AstraZeneca COVID-19 vaccination: https://ncbi.nlm.nih.gov/34753210/.
- COVID-19 vaccine in patients with hypercoagulability disorders: a clinical perspective: https://pubmed.ncbi.nlm.nih.gov/34786893/
- Spectrum of neuroimaging findings in post-CoVID-19 vaccination: a case series and review of the literature: https://pubmed.ncbi.nlm.nih.gov/34842783/
- Increased risk of urticaria/angioedema after BNT162b2 mRNA COVID-19 vaccination in health care workers taking ACE inhibitors: https://pubmed.ncbi.nlm.nih.gov/ 34579248/
- Central retinal vein occlusion after vaccination with SARS-CoV-2 mRNA: case report: https://pubmed.ncbi.nlm.nih.gov/34571653/.
- Idiopathic external jugular vein thrombophlebitis after coronavirus disease vaccination (COVID-19): https://pubmed.ncbi.nlm.nih.gov/33624509/.
- Squamous cell carcinoma of the lung with hemoptysis following vaccination with tozinameran (BNT162b2, Pfizer-BioNTech): https://pubmed.ncbi.nlm.nih.gov/ 34612003/
- Chest pain with abnormal electrocardiogram redevelopment after injection of COVID-19 vaccinemanufactured by Moderna: https://pubmed.ncbi.nlm.nih.gov/ 34866106/
- Reporting of acute inflammatory neuropathies with COVID-19 vaccines: subgroup disproportionalityanalysis in VigiBase: https://pubmed.ncbi.nlm.nih.gov/34579259/
- Brain death in a vaccinated patient with COVID-19 infection: https://ncbi.nlm.nih.gov/34656887/
- Kounis syndrome type 1 induced by inactivated SARS-COV-2 vaccine: https://ncbi.nlm.nih.gov/34148772/
- Rapid progression of angioimmunoblastic T-cell lymphoma after BNT162b2 mRNA booster vaccination:case report: https://frontiersin.org/articles/10.3389/fmed. 2021.798095/
- Gastroparesis after Pfizer-BioNTech COVID-19 vaccination: https://ncbi.nlm.nih.gov/34187985/
- Colaneri, , De Filippo, M., Licari, A., Marseglia, A., Maiocchi, L., Ricciardi, A., . . . Bruno, R. (2021). COVID vaccination and asthma exacerbation: might there be a link? Int J Infect Dis, 112, 243-246. doi:10.1016/j.ijid.2021.09.026. https:// www.ncbi.nlm.nih.gov/pubmed/34547487
- Dimopoulou, D., Spyridis, N., Vartzelis, G., Tsolia, M. N., & Maritsi, D. N. (2021). Safety and tolerability of the COVID-19 mRNA-vaccine in adolescents with juvenile idiopathic arthritis on treatment with TNF-inhibitors. Arthritis doi:10.1002/ art.41977. https://www.ncbi.nlm.nih.gov/pubmed/34492161
- Hause, M., Gee, J., Baggs, J., Abara, W. E., Marquez, P., Thompson, D., . . . Shay,
- K. (2021). COVID-19 Vaccine Safety in Adolescents Aged 12-17 Years – United States, December 14,2020-July 16, 2021. MMWR Morb Mortal Wkly Rep, 70(31),
1053-1058. doi:10.15585/mmwr.mm7031e1. https://www.ncbi.nlm.nih.gov/pubmed/
34351881
- Shay, D. K., Gee, J., Su, J. R., Myers, T. R., Marquez, P., Liu, R., . . . Shimabukuro, T.
- (2021). Safety Monitoring of the Janssen (Johnson & Johnson) COVID-19 Vaccine – United States, March-April 2021. MMWR Morb Mortal Wkly Rep, 70(18), 680-684. doi:10.15585/mmwr.mm7018e2. https://www.ncbi.nlm.nih.gov/pubmed/33956784
- Acute myocardial injury after COVID-19 vaccination: a case report and review of current evidence from the Vaccine Adverse Event Reporting System database: https://pubmed.ncbi.nlm.nih.gov/34219532/
- Deb, , Abdelmalek, J., Iwuji, K., & Nugent, K. (2021). Acute Myocardial Injury Following COVID-19 Vaccination: A Case Report and Review of Current Evidence from Vaccine Adverse Events ReportingSystem Database. J Prim Care Community Health, 12, 21501327211029230. doi:10.1177/21501327211029230. https://
www.ncbi.nlm.nih.gov/pubmed/34219532
- Furer, , Eviatar, T., Zisman, D., Peleg, H., Paran, D., Levartovsky, D., . . . Elkayam,
- (2021). Immunogenicity and safety of the BNT162b2 mRNA COVID-19 vaccine in adult patients with autoimmune inflammatory rheumatic diseases and in the general population: a multicentre study. Ann Rheum Dis, 80(10), 1330-1338. doi:10.1136/ annrheumdis-2021-220647. https://www.ncbi.nlm.nih.gov/pubmed/34127481
- Neurological autoimmune diseases after SARS-CoV-2 vaccination: a case series:https://pubmed.ncbi.nlm.nih.gov/34668274/.
- Clinical and pathologic correlates of skin reactions to COVID-19 vaccine, including V- REPP: a registry-based study: https://www.sciencedirect.com/science/article/pii/ S0190962221024427
- Varicella zoster virus and herpes simplex virus reactivation after vaccination with COVID-19: review of 40 cases in an international dermatologic registry: https:// pubmed.ncbi.nlm.nih.gov/34487581/
Può interessarti anche: Austria: cosa vuole sapere la Corte Costituzionale dal ministero della salute. Le domande scomode
Seguici su Facebook https://www.facebook.com/presskit.it
Seguici su Telegram https://t.me/presskit